Leukemia Treatment Articles & Analysis: Older
11 articles found
A major breakthrough occurred with the FDA approval of Venetoclax (ABT-199), a BCL2-selective inhibitor used in the treatment of chronic lymphocytic leukemia (CLL). Venetoclax works by mimicking BH3-only proteins, thereby displacing pro-survival BCL2 proteins and triggering apoptosis in cancer cells. In 2020, a study published in the New England Journal of ...
Enter immunotherapy - a groundbreaking approach that is not only redefining leukemia treatment but also offering new hope as a potent solution provider for this challenging condition. Immunotherapy in Leukemia Treatment Immunotherapy represents a paradigm shift in the treatment of leukemia. ...
Currently, 9 ADCs have been approved, of which 5 are used for hematoma and 4 for solid tumor treatment. After more than 20 years of development, ADC biopharmaceuticals are becoming the main force in the treatment of cancer. ...
The real ADC was not approved for marketing by the US FDA until 2000, that is, Wyeth's gemtuzumab, in which the antibody is a recombinant humanized CD33 monoclonal antibody conjugated with the cytotoxin calicheamicin for the treatment of acute myeloid leukemia. However, through a large number of clinical studies, it was found that it could not improve the ...
At present, chimeric antigen receptor T cell therapy (CAR-T) has made significant achievements in the treatment of specific hematological cancers, allowing patients with relapsed/refractory disease to survive longer and become healthier, but in clinical studies, cell therapy has actually not been successful in the treatment of patients with solid tumors, in part ...
Sandwiching the wobble protein between two other layers allowed scientists to get the most detailed picture of a protein that is key to the spread of acute myeloid leukemia. Acute myeloid leukemia (AML) afflicts more than 20,000 Americans each year, killing more than 11,000 of them, according to the American Cancer Institute. ...
It's worth noting, however, that this treatment has very limited use. Stem cell transplantation is fraught with danger, and if used solely to treat AIDS, it is no better than antiviral drugs. It can only be considered for HIV eradication by chance if the patient requires blood stem cells for other diseases—in this case, the patient was transplanted for acute ...
Mylotarg, Besponsa, and first-generation cleavable linkers In 2000, the FDA authorized Mylotarg for the treatment of acute myeloid leukemia (AML). It consists of a cleavable linker containing a hydrazone bond that connects kacinomycin to gemtuzumab (a mutant anti-CD33 IgG4 subtype monoclonal antibody). ...
The FDA approved CAR-T cell therapy, also known as chimeric antigen receptor T-cell therapy, as the first CAR-T cell therapy to be marketed for the treatment of leukemia in 2017, ushering in a new age of CAR-T cell therapy. ...
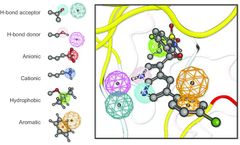
STI-571 is a classic tyrosine kinase inhibitor for the treatment of chronic myeloid leukemia. Among different tyrosine kinases (ABL, c-KIT, SYK), the compound exhibits different binding conformations. 1. ...
In 1958, Mathe first conjugated anti-mouse antibody with methotrexate for the treatment of leukemia. Because of the problems of immunogenicity and antibody preparation, ADC drugs stopped decades ago, until the emergence of monoclonal antibodies in 1975, and later the emergence of humanized antibodies. In 2000, the first antibody-conjugated drug was approved by ...