- Home
- Companies
- Centinel Spine
- Products
Centinel Spine products
Lumbar
Model prodisc L - Anterior Lumbar Total Disc Replacement
The most studied and clinically-proven total disc replacement technology in the world is now the only total disc replacement system in the U.S. approved for two-level use in the lumbar spine.
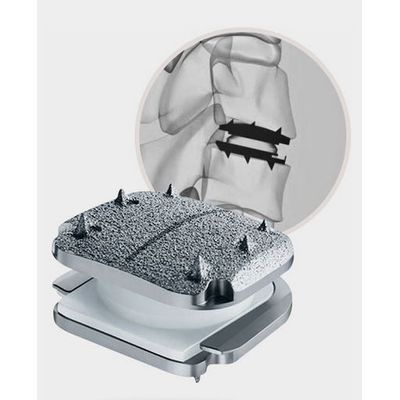
Stalif - Model M Flx - Anterior Lumbar Integrated Interbody System
3D-printed porous titanium lumbar interbody with 2-level clearance!
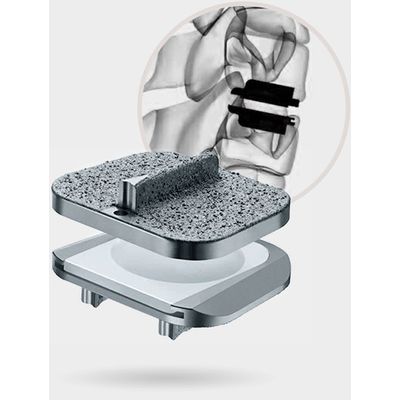
Stalif - Model M Ti - No-Profile Anterior Lumbar Integrated Interbody System
Sixth generation STALIF® technology, STALIF M-Ti™ incorporates a novel, circumferential, titanium surface, PEEK Integrated Interbody™ design that is raising the Gold Standard of lumbar interbody fusion.
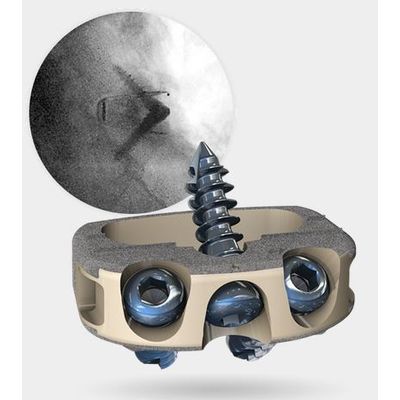
Model STALIF L - Integrated Interbody™ System
Clinical Applications: Addressing Crucial Clinical Needs. STALIF L™ continues the 25+ year STALIF® heritage of innovation. Developed with prominent spine surgeons to address an unmet need, STALIF L meets the demands and complexity of minimally invasive lumbar lateral interbody fusion procedures. Surgical Ease: Integrated self-tapping, self-drilling screws simplify implant immobilization with optimized screw angulation to provide strength and stability without interfering with posterior hardware. Segmental Alignment & Stability: Recreate Segmental Alignment and Stability, Combining integrated fixation with implant lengths ranging from 45mm to 55mm, STALIF L implants seat across the entire apophyseal ring to recreate segmental alignment and stability. Restore Sagittal Balance: With a variety of footprint and height options available in 0 and 12 degrees of lordosis, STALIF L provides many options to restore anatomical alignment.
Cervical
Prodisc - Model C - Anterior Cervical Total Disc Replacement
The most studied and clinically-proven total disc replacement technology in the world.
Prodisc - Model C - Vivo - Anterior Cervical Total Disc Replacement
The most studied and clinically-proven total disc replacement technology in the world.
Model Prodisc C Nova - Anterior Cervical Total Disc Replacement
The most studied and clinically-proven total disc replacement technology in the world. Determined Safe & Effective for SCDD: The prodisc C Nova Total Disc Replacement is intended to replace a diseased and/or degenerated intervertebral disc of the cervical spine in patients with symptomatic cervical disc disease (SCDD). The prodisc C Nova Total Disc Replacement surgery is intended to: Significantly reduce pain by allowing for the removal of the diseased disc, Restore biomechanical stability, Restore normal disc height, Provide the potential for motion at the affected vertebral segment.
Model STALIF C FLX - Anterior Cervical Integrated Interbody System
3D-printed porous titanium cervical interbody with 2-level clearance! lexible: FLX implants include a solid exoskeleton for added strength, along with a proprietary lattice scaffold designed to have a stiffness similar to bone. Reduced device stiffness is known to minimize stress-shielding and enhance the fusion process. Lucent: FLX implants include porous radiolucent sections with optimized density to reduce imaging artifacts—enhancing intra-operative visualization & post-operative fusion assessment compared to solid titanium implants. MatriX: FLX implants include a FUSE-THRU™ osteoconductive trabecular scaffold, bioengineered to allow the potential for bony on-growth and in-growth throughout the implant. This unique porous structure contains micro, macro, and nano structural characteristics designed to promote the fusion healing process by influencing cellular events related to the production of new bone.
Model STALIF C-Ti - No-Profile® Anterior Cervical Integrated Interbody System
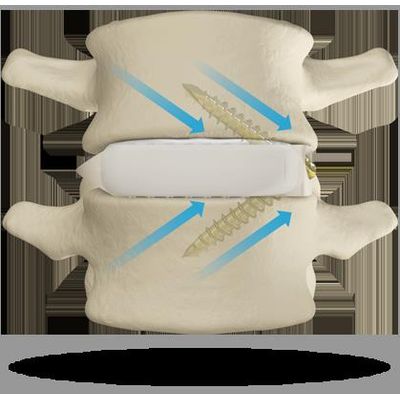
Model ALTOS PCT - Evolving the Gold Standard in Cervical Spinal Fusion Procedures
Introducing the First FDA cleared posterior cervical stabilization system specifically designed for use in either the cervical lateral masses or cervical-thoracic pedicles. Delivering the Results Surgeons Demand: Delivering gold standard clinical outcomes with a simple, comprehensive posterior cervical-thoracic stabilization system. Minimize tissue irritation for enhanced patient comfort with a low-profile screw and rod system, Simple rod placement resulting from friction stabilized tulip head, Streamlined surgical procedure performed with simple, low-volume instrument set