Integer Holdings Corporation products
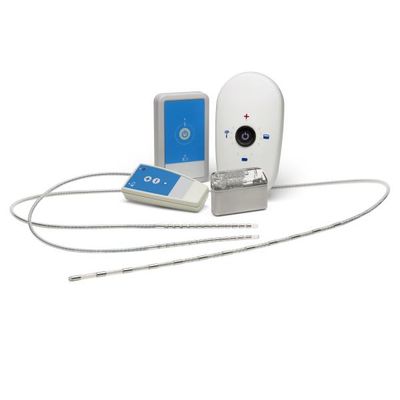
Integer - Model Class III - Medical Device Systems
As a Class III Medical Device manufacturer, our OEM partners trust us to custom design, develop and manufacture their implantable pulse generators (IPG) and lead systems. We honor that trust with a focus on accelerating time to market and reducing program risk and commercialization costs. IPGs are an integral part of our history, in the last 40 of our 70+ years, we have designed and developed over 40+ custom IPG systems for customers. Our unique combination of deep expertise, proven technology modules, device platforms and vertically integrated supply chain allow us to deliver complex custom IPG systems, while de-risking program execution and accelerating time to market.
Introducers
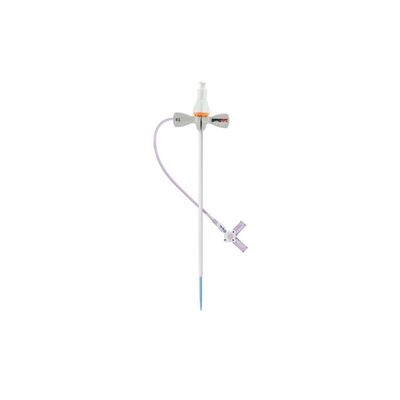
OSCOR Adelante SafeSheath - Model II - Hemostatic Peel Away Introducer with Side Port
Introducing the Adelante® SafeSheath® II Hemostatic Peel Away Introducer System for Vascular Access. This generation peel away introducer features the latest lubricated hemostatic valve membrane that provides low insertion forces during procedures. It also includes a side port with a 3-way stopcock that provides a convenient means of aspirating and flushing the introducer.
Adelante SafeSheath - Model Ultra Lite - Hemostatic Valve Peel Away System
The Adelante Safesheath Ultra Lite Peel Away Introducer System, features a specially engineered valve membrane that allows for reduced insertion forces during procedures.
OSCOR Adelante - Advanced Peel-Away Introducer
The Adelante® Peel Away Introducer System is one of the most advanced introducers available for placement of permanent pacing leads and catheters. It offers exceptional insertion, smooth lead movement, high kink resistance and exceptional peeling performance even in complex conditions.
Batteries
Integer - Implantable Battery
Our implantable batteries are built upon unparalleled quality standards delivering safe and dependable power to your medical device. We provide standard options for Primary (Non-rechargeable) and Secondary (Rechargeable) cells, as well as customized power source solutions that are designed and manufactured to meet customer unique requirements.
Integer - Portable Battery Packs
Our in-depth knowledge of battery cell technologies and battery pack design, as well as medical device and regulatory expertise, drives us to be the premier portable power source to our customers. Whether you are seeking to build and power a medical device or simply seeking portable battery packs, our team of scientists and engineers will help you navigate the complexities of the numerous battery chemistries and their performance and safety to select the best option for your needs. A dedicated project team will then work closely with your team to develop and manufacture the complete solution leveraging our global supply chain. With Integer, you have:
Capacitors
Greatbatch Medical - Model QCAPS - High Voltage Tantalum Capacitors for Tachycardia Device
The Greatbatch Medical™ QCAPS™ high voltage tantalum capacitors provide fast, consistent charge times for tachycardia therapy, the first time and every time. When paired with QHR® batteries, QCAPS capacitors provide the smallest, longest-lived, highest-energy power solutions for today’s tachycardia devices.
Catheters
Integer - Ablation Catheters
Integer - Coronary CTO Microcatheters
We provide a full range of coronary CTO microcatheter solutions to support every phase of the product life cycle. Design and development of coronary CTO microcatheters. Transfer of products and processes from one site to another to support product-line transfer, line duplication, low-cost manufacturing, and prototype-to-production migration. Manufacture of standard and custom cardiovascular medical-device components, including polyimide-coated wire, high-spring constant coils, braided shafts, polyimide tubing, braided multi-lumen extrusions, PT electrodes and marker bands, welded electrode assemblies, coated stainless wire-welded pull cables, thermocouples, micro-machined ablation tips, micro-molding, and machined and molded handles. Design for manufacturability (DFM) to assure that final products can be repeatedly manufactured while meeting quality, reliability, and performance requirements.
Integer - Diagnostic Mapping Catheters
We provide a full range of solutions to support every phase of the product life cycle of diagnostic mapping catheters. Design and development of complex diagnostic mapping catheters including diagnostic EP catheters, high-density mapping catheters and ICE catheters. Transfer of products and processes from one site to another to support product-line transfer, line duplication, low-cost manufacturing, and prototype-to-production migration. Manufacture of completed diagnostic mapping catheters as well as custom medical device components, including polyimide-coated wire, high-spring constant coils, steerable braided shafts, polyimide tubing, braided multi-lumen extrusions, PT electrodes and marker bands, welded electrode assemblies, coated stainless wire-welded pull cables, thermocouples, micro-molding, and machined and molded handles.