- Home
- Companies
- Co-Diagnostics, Inc.
- Products
- Logix Smart - COVID-19Test Kit
Logix Smart - COVID-19Test Kit
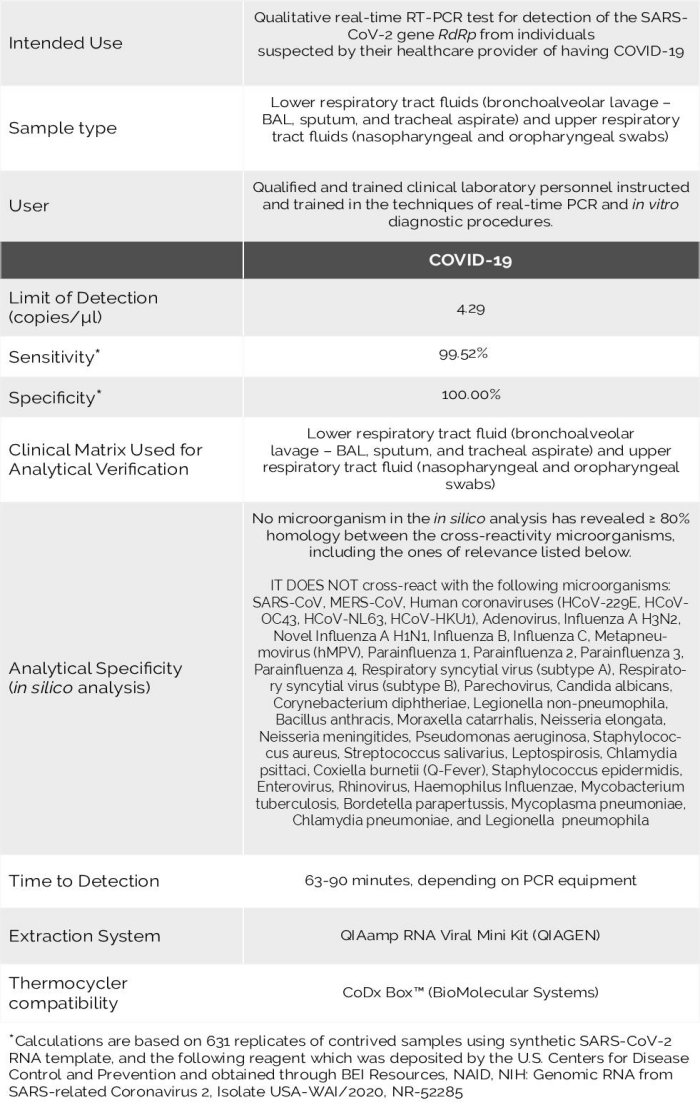
The Logix Smart™ Coronavirus Disease 2019 (COVID-19) Test kit is an in vitro diagnostic test that uses our patented CoPrimer™ technology for the qualitative detection of the RNA from SARS-CoV-2 coronavirus (COVID-19). The test operates using a single step real-time reverse transcriptase polymerase chain reaction (RT-PCR) process in lower respiratory tract fluids (e.g. bronchoalveolar lavage, sputum, tracheal aspirate), and upper respiratory tract fluids (e.g. nasopharyngeal and oropharyngeal swabs) from patients who meet the clinical criteria (e.g. signs and symptoms) for coronavirus disease 2019 (COVID-19) as established by WHO (WHO, 2020) and the US CDC (CDC, 2020) (e.g. fever, cough, shortness of breath, travel history to China).
Click here to read the Company’s regulatory bulletin on the ability of our suite of Logix Smart COVID-19 diagnostics to detect all known strains of SARS-CoV-2.
- Ready-to-use Master Mix, complete with RNaseP internal positive control to verify sample quality
- Positive Control (PC), to verify the performance of the master mix
- Nuclease-Free Water as a negative control, to verify master mix is free from contamination
COVID-19 is a contagious respiratory illness caused by the SARS-CoV-2 virus, a relatively new virus in humans. COVID-19 can present with a mild to severe illness, although some people infected with COVID-19 may have no symptoms at all. Older adults and people of any age who have underlying medical conditions have a higher risk of severe illness from COVID-19. Serious outcomes of COVID-19 include hospitalization and death. The SARS-CoV-2 virus can be spread to others not just while one is sick, but even before a person shows signs or symptoms of being sick (e.g., fever, coughing, difficulty breathing, etc.). A full list of symptoms of COVID-19 can be found at the following link:
The Logix Smart Coronavirus Disease 2019 (COVID-19) test is a real-time RT-PCR test using a proprietary technology called CoPrimers™ (Satterfield, 2014)(Poritz & Ririe, 2014) intended for the qualitative detection of nucleic acid from the SARS-CoV-2 in lower respiratory tract fluids (e.g. bronchoalveolar lavage, sputum, tracheal aspirate), and upper respiratory tract fluids (e.g. nasopharyngeal and oropharyngeal swabs) from individuals with signs and symptoms of infection who are suspected of having COVID-19. The test is available to laboratories certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA), 42 U.S.C. §263a, to perform high complexity tests, or by similarly qualified non-U.S. laboratories.
Negative results do not preclude SARS-CoV-2 infection and should not be used as the sole basis for patient management decisions. Negative results must be combined with clinical observations, patient history, and epidemiological information.
The Logix Smart Coronavirus Disease 2019 (COVID-19) is intended for use by trained clinical laboratory personnel specifically instructed and trained in the techniques of real-time PCR and in vitro diagnostic procedures. The Logix Smart Coronavirus Disease 2019 (COVID-19) test is only for use under the Food and Drug Administration’s Emergency Use Authorization.
Poritz, M., & Ririe, K. (2014, Mar). Getting things backwards to prevent primer dimers. Journal of Molecular Diagnosis, 159-62. doi:10.1016/j.jmoldx.2014.01.001
Satterfield, B. (2014, Mar). Cooperative primers: 2.5 million-fold improvement in the reduction of nonspecific amplification. Journal of Molecular Diagnosis, 163-73. doi:10.1016/j.jmoldx.2013.10.004
Instructions for Use can be found under the Resources tab.