- Infusion Therapy - IV Solutions, Pre-Mixed Drugs, and Irrigation Solutions - Accessories for IV Containers
- Infusion Therapy - IV Solutions, Pre-Mixed Drugs, and Irrigation Solutions - General Purpose IV Solutions
- Infusion Therapy - IV Solutions, Pre-Mixed Drugs, and Irrigation Solutions - Multi-Electrolytes
- Regional Anesthesia and Pain Management
- Dextrose and Sodium Chloride Injections
B. Braun Medical Inc. products
Infusion Therapy - IV Solutions, Pre-Mixed Drugs, and Irrigation Solutions - Accessories for IV Containers
B-Braun - Additive Cap (Non-Sterile) for PAB Bags
Components not made with natural rubber, latex, PVC, or DEHP.
Infusion Therapy - IV Solutions, Pre-Mixed Drugs, and Irrigation Solutions - General Purpose IV Solutions
B-Braun - Model EXCEL IV Container - 5% Dextrose Injections
Available in 250 mL, 500 mL and 1000 mL EXCEL IV containers and 100 mL and 150 mL PAB Partial Additive containers. Dextrose Injections USP are sterile, nonpyrogenic and contain no bacteriostatic or antimicrobial agents. These products are intended for intravenous administration. These intravenous solutions are indicated for use in adults and pediatric patients as sources of calories and water for hydration.
B-Braun - Model 1000 mL - 10% Dextrose Injection USP
Dextrose Injections USP are sterile, nonpyrogenic and contain no bacteriostatic or antimicrobial agents. These products are intended for intravenous administration. These intravenous solutions are indicated for use in adults and pediatric patients as sources of calories and water for hydration.
Infusion Therapy - IV Solutions, Pre-Mixed Drugs, and Irrigation Solutions - Multi-Electrolytes
Isolyte - Model P - Multi-Electrolyte Injection
Isolyte P in 5% Dextrose is sterile, nonpyrogenic, and contains no bacteriostatic or antimicrobial agents or added buffers. This product is intended for intravenous administration. This solution is indicated for use in adults as a source of electrolytes, calories and water for hydration, and as an alkalinizing agent. Not made with latex, PVC or DEHP.
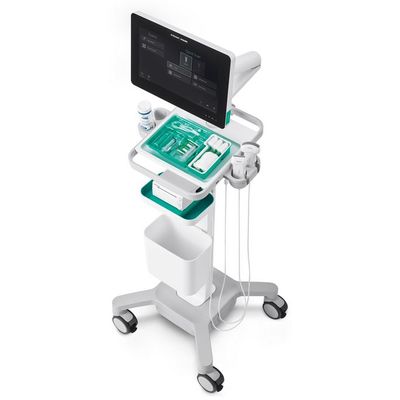
Regional Anesthesia and Pain Management
Xperius - Ultrasound System
B. Braun Melsungen AG and Royal Philips have formed a multi-year strategic alliance to innovate ultrasound-guided regional anesthesia – a rapidly growing alternative to general anesthesia – and vascular access. B. Braun, the global leader in regional anesthesia and pain management, and Philips, a global leader in image guided therapy solutions, are leveraging their combined clinical expertise and R&D capabilities, as well as sales and service channels, to jointly develop and commercialize solutions to support anesthesiologists and hospitals in critical areas of regional anesthesia. The alliance will also focus on vascular access procedures, such as those used to insert catheters into deeply seated veins as part of a catheter-based treatment.
Dextrose and Sodium Chloride Injections
EXCEL IV - Model L6080-00 - 3.3% Dextrose and 0.30% Sodium Chloride Injection USP, 1000 mL
Dextrose and Sodium Chloride Injections USP are sterile, nonpyrogenic and contain no bacteriostatic or antimicrobial agents. These products are intended for intravenous administration. These intravenous solutions are indicated for use in adults and pediatric patients as sources of electrolytes, calories and water for hydration. Not made with latex, PVC or DEHP.
EXCEL IV - 5% Dextrose and 0.9% Sodium Chloride Injection USP, 1000 mL
Dextrose and Sodium Chloride Injections USP are sterile, nonpyrogenic and contain no bacteriostatic or antimicrobial agents. These products are intended for intravenous administration. These intravenous solutions are indicated for use in adults and pediatric patients as sources of electrolytes, calories and water for hydration. Not made with latex, PVC or DEHP.
EXCEL IV - 5% Dextrose and 0.9% Sodium Chloride Injection USP, 500 mL
Dextrose and Sodium Chloride Injections USP are sterile, nonpyrogenic and contain no bacteriostatic or antimicrobial agents. These products are intended for intravenous administration. These intravenous solutions are indicated for use in adults and pediatric patients as sources of electrolytes, calories and water for hydration. Not made with latex, PVC or DEHP.
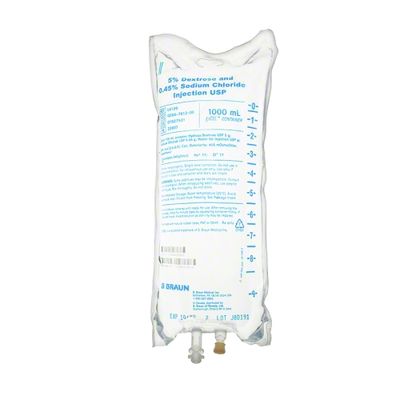
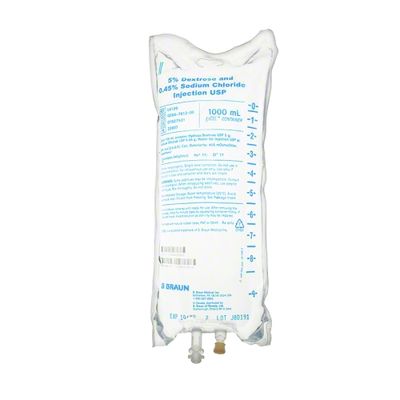
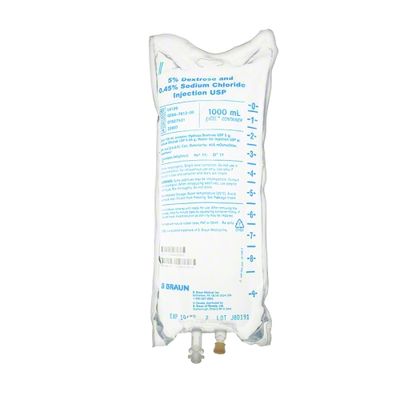
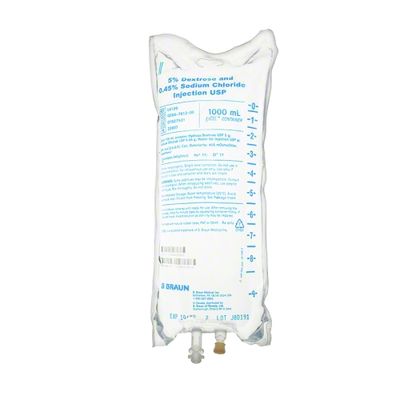
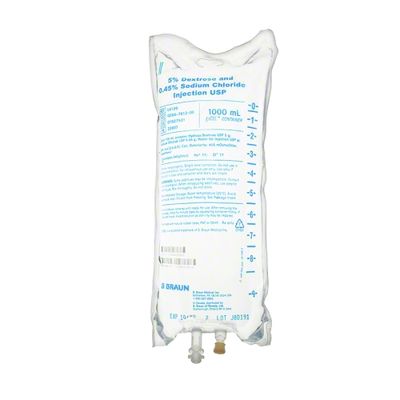
EXCEL IV - 5% Dextrose and 0.45% Sodium Chloride Injection USP, 1000 mL
Dextrose and Sodium Chloride Injections USP are sterile, nonpyrogenic and contain no bacteriostatic or antimicrobial agents. These products are intended for intravenous administration. These intravenous solutions are indicated for use in adults and pediatric patients as sources of electrolytes, calories and water for hydration. Not made with latex, PVC or DEHP. Specific Gravity: 1.020
EXCEL IV - 5% Dextrose and 0.45% Sodium Chloride Injection USP, 500 mL
Dextrose and Sodium Chloride Injections USP are sterile, nonpyrogenic and contain no bacteriostatic or antimicrobial agents. These products are intended for intravenous administration. These intravenous solutions are indicated for use in adults and pediatric patients as sources of electrolytes, calories and water for hydration. Not made with latex, PVC or DEHP.