bioactiva diagnostica GmbH products
Medical Equipment - Urology
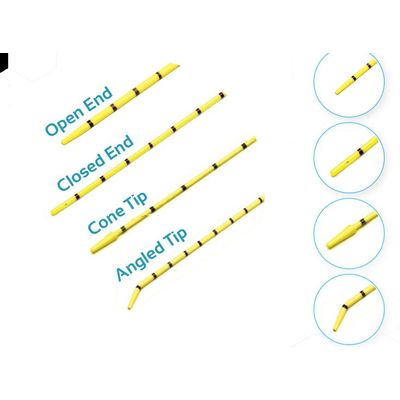
Model BUCP003070B1 - Ureteral Catheter
Radiopaque for better visualization, 1 cm marking helps in catheter positioning and measuring the length of ureter, Detachable hub for scope insertion, Size (in Fr): 3, 4, 5, 6 & 7, Length (in cm): 70.
Model BPCP006030B1 - PCN Catheter
The pigtail tip configuration helps in retention of the distal tip in the renal pelvis, Eye holes for better drainage, Hydrophilic coating for easy insertion, Size (in Fr): 6, 7, 8, 9, 10, 12, 14 & 16, Length (in cm): 30
Model BMCP008030B1 - Malecot Catheter
The Malecot flower design is ideally suitable for the drainage of thick viscous fluids, Integral flower is designed to match the catheter size. This reduces trauma during insertion, Size (in Fr) 8, 10, 12, 14, 16, 18, 20 & 22, Length (in cm) 30.
Veterinary
Model 11-263-001 - Intray® Sab-Fungid™
Intended Use:- InTray® SAB-FungID™ contains Sabouraud’s dextrose agar, a nonselective medium used to aid in the detection of dermatophyte fungi Nfrom clinical specimens with mixed microbiota. Expiry date:- 10-12 Months, Certification:- CE IVD, Company:- Biomed Diagnostics, Inc., Storage condition:- 2 - 8° C, Materials Provided:- InTray SAB-FungID test(s), Product Number:- 11-263-001, Manufacturer:- Biomed Diagnostics, Inc., Order:- Request Quote, Certification:- CE IVD
Model 11-283-001 - Intray® Sab-Fungid™ W/Cc
Intended Use:- InTray® SAB-FungID™ w/ CC contains Sabouraud’s dextrose agar with chloramphenicol and cycloheximide, a selective medium used to aid in the detection of dermatophyte fungi from clinical specimens with mixed microbiota. Expiry date:- 10-12 Months, Certification:- CE IVD, Company:- Biomed Diagnostics, Inc., Storage condition:- 2 - 8° C, Materials Provided:- InTray SAB-FungID w/ CC test(s), Product Number:- 11-283-001, Manufacturer:- Biomed Diagnostics, Inc., Order:- Request Quote, Certification:- CE IVD
Immunoassays
Model ADVG0010BA - Adenovirus IgG
Intended Use:- The Adenovirus IgG ELISA is intended for the qualitative determination of IgG class antibodies against Adenovirus in human serum or plasma (citrate, heparin). Expiry date:- 10-12 Months. Certification:- CE IVD.Company:- bioactiva diagnostica GmbH. Storage condition:- 2 - 8 °C. Running time:- 105 min. Materials Provided:- Microtiterplate, IgG Sample Dilution Buffer, Stop Solution, Washing Buffer, Conjugate, TMB Substrate Solution, Positive Control, Cut-off Control, Negative Control, 1 Cover foil, 1 Instruction for use (IFU), 1 Plate layout, Product Number:- ADVG0010BA. Manufacturer:- bioactiva diagnostic. Order:- Buy Now. Certification:- CE IVD
Model ADVM0010BA - Adenovirus IgM
Intended Use:- The Adenovirus IgM ELISA is intended for the qualitative determination of IgM class antibodies against Adenovirus in human serum or plasma (citrate, heparin). Expiry date:- 10-12 Months. Certification:- CE IVD. Company:- bioactiva diagnostica GmbH. Storage condition:- 2 - 8 °C. Running time:- 105 min. Materials Provided:- Microtiterplate, IgM Sample Dilution Buffer, Stop Solution, Washing Buffer, Conjugate, TMB Substrate Solution, Positive Control, Cut-off Control, Negative Control , 1 Cover foil , 1 Instruction for use (IFU) , 1 Plate layout. Product Number:- ADVM0010BA. Manufacturer:- bioactiva diagnostic. Order:- Buy Now. Certification:- CE IVD
Model CHIG0590 - Chikungunya Virus IgG Capture
Intended Use:- The Chikungunya Virus IgG capture ELISA is intended for the qualitative determination of IgG class antibodies against Chikungunya Virus in human serum or plasma (citrate, heparin). Expiry date:- 10-12 Months. Certification:- CE IVD. Company:- NovaTec Immundiagnostica GmbH. Storage condition:- 2 - 8 °C. Running time:- 165 min. Materials Provided:- Microtiterplate, Sample Dilution Buffer, Stop Solution, Washing Buffer (20x conc.), Antigen, Lyophilized, Antibody Solution, Conjugate, TMB Substrate Solution, Positive Control, Cut-off Control, Negative Control, 1 Cover foil, 1 Instruction for use (IFU) , 1 Plate layout. Product Number:- CHIG0590. Manufacturer:- NovaTec Immundiagnostica. Order:- Request Quote. Certification:- CE IVD
Model CHIM0590 - Chikungunya Virus IgM µ-Capture
Intended Use:- The Chikungunya Virus IgM μ-capture ELISA is intended for the qualitative determination of IgM class antibodies against Chikungunya Virus in human serum or plasma (citrate, heparin). Expiry date:- 10-12 Months. Certification:- CE IVD. Company:- NovaTec Immundiagnostica GmbH. Storage condition:- 2 - 8°C. Materials Provided:- Microtiterplate, Sample Dilution Buffer, Stop Solution, Washing Buffer (20x conc.), Antigen Solution, lyophilized, Antibody Solution, Conjugate, TMB Substrate Solution, Positive Control, Cut-off Control, Negative Control, 1 Cover foil, 1 Instruction for use (IFU) , 1 Plate layout. Product Number:- CHIM0590. Manufacturer:- NovaTec Immundiagnostica. Order:- Request Quote. Certification:- CE IVD.