Xtant Medical products
Biologics
Xtant 3Demin - Demineralized Cortical Bone Fibers
Xtant’s 3Demin Technology utilizes demineralized cortical bone fibers that are entangled and shaped into sizes engineered to complement specific surgical applications. This unique process creates an interconnected graft material that contains BMPs and other growth factors necessary for the promotion of new bone formation. 3Demin allografts are flexible upon hydration and each allograft has a sterility assurance level of 10-6 via low-dose gamma sterilization. 3Demin allografts are also available as loose cortical fibers in three volume options.
Matriform - Model Si - Synthetic Grafts
Matriform Si was developed to resemble the composition and porous structure of natural human bone. Comprised of 96% pure phase β-TCP granules, 4% silicate and collagen, Matriform Si provides the ideal biomimetic scaffold for spinal fusion procedures. The flexible strip offers excellent handling and shape memory ensuring direct contact with the surface of healthy bone.
OsteoMax - Demineralized Bone Matrix Putty
Derived from 100% human bone – maximum allograft content; no extraneous carriers that may impede bone healing; Osteoinductive potential – validated for positive osteoinductive capacity; Osteoconductive – DBM provides an ideal scaffold that directs and supports bone formation; Excellent handling characteristics – retains shape and resists irrigation and migration; Convenient and ready to use – stores at room temperature and ready for immediate use; Sterility assurance level (SAL) 10-6 – terminal sterilization ensures the highest level of patient safety.
OsteoSelect - Osteoinductivity Graft
OsteoSelect was engineered for superior performance. The graft does not adhere to gloves yet maintains its placement in the surgical environment. Osteoinductivity of sterile final product is assessed in vivo. In this challenging model, every lot tested to date has consistently demonstrated an osteoinductive response. This performance validates Xtant Medical’s expertise in DBM processing.
OsteoVive - Model Plus - Osteogenic Potential-Proprietary Processing System
Osteogenic Potential- Proprietary processing preserves adult mesenchymal stem cells while removing red and white blood cells. Osteoinductive Potential- Cortical fiber demineralized bone matrix provides a high quality osteoinductive signal. Additional growth factors are captured from bone lining cells and restored back into the product.* Osteoconductive- The cortical fiber demineralized bone matrix and cancellous bone creates a robust scaffold. Ease of Use- 10 minute thaw time in a room temperature water bath. OsteoVive Plus is DMSO free and immediately implantable upon thawing in a ready-to-use syringe.
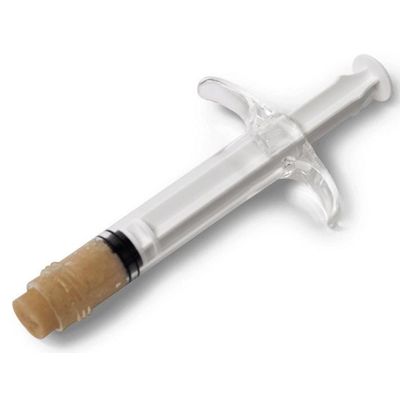
SimpliMix - Graft Delivery Device
The SimpliMix™ Graft Delivery Device is designed for targeted delivery of hydrated allograft, autograft, or synthetic bone graft materials to an orthopedic surgical site, while maximizing material utilization.
SportsMed - Sports Medicine Allografts
Sports Medicine allografts are an excellent replacement option for deficient or torn cruciate ligament reconstructions and revisions. Benefits include consistent, superior quality and precise measurements. As with all of Xtant Medical’s allografts, safety and efficacy is an absolute priority. All tendons are distributed sterile via low-dose gamma irradiation.
Fixation
Aranax - Anterior Cervical Plate and Screws
An anterior cervical fixation system, Aranax incorporates a lower profile plate to minimize soft tissue disruption, increased screw angulation to enhance bone purchase, and an integrated locking mechanism to help prevent screw back-out. All implants are sterile packaged for product quality.
Atrix-C Union - Allograft Cervical Interbody Spacer
The Atrix-C Union allograft cervical interbody spacer provides an osteoconductive matrix creating an optimal environment for bone growth and fusion. Atrix-C Union allografts are available in three footprints and eight heights to accommodate a wide range of patient anatomy while providing a steady platform for fusion.
Axle - Interspinous Fusion System
The Axle Interspinous Fusion System is an internal fixation device for posterior spinal surgery in the non-cervical spine (T1-S1 inclusive). It is a minimally invasive, modular Interspinous fusion system with straightforward instrumentation. The Axle Interspinous Fusion System is designed to provide spinal stability for lumbar fusion procedures, including the treatment of degenerative disc disease, spinal tumors and trauma.