Refine by
Biopharmaceutical Drug Articles & Analysis
10 articles found
Host cell protein (HCP) residual detection refers to the testing of proteins that may remain in the final product and originate from the production cell line (i.e., the "host") during the biopharmaceutical production process. This testing is crucial because these host cell proteins (HCPs) may impact the safety, efficacy, and quality of drug products.In the fields ...
Poor water solubility can limit the bioavailability and therapeutic efficacy of many drugs. PEG derivatives, with their hydrophilic nature, enhance the solubility and stability of hydrophobic drugs. ...
Biopharmaceuticals and personalized medicine are hot fields in the development of the pharmaceutical industry today. ...
Oncology drug development makes up almost half of the annual R&D expenditure of biopharmaceutical companies. Only 10% of drugs advanced to the clinical stage are successfully brought to market, with half of all failures attributed to lack of clinical efficacy. ...
Genomic engineering in cell lines is a versatile tool for studying gene function, designing diseases models, biopharmaceutical research, drug discovery and many other applications. CRISPR (Clustered Regulatory Interspaced Short Palindromic Repeats)/Cas9 systems is a newly developed yet the most popular method for genome editing. ...
Biopharma PEG, a leading innovator in polyethylene glycol (PEG) derivatives, proudly introduces its featured Azide PEG series, which can be used in drug development, click chemistry, ADCs (Antibody Drug Conjugates), PROTACs (Proteolysis Targeting Chimeras), and beyond. This Azide PEG features monodispersed PEGs such as N3-PEG4-OH, N3-PEG3-COOH, N3-PEG11-NH2, N3-PEG3-NHS ester, ...
Hydrogels are composed of hydrophilic polymers, whose three-dimensional network structure can not only absorb a large amount of water, but also be used to carry drugs. Hydrogels prepared with suitable materials have the characteristics of high biocompatibility, mechanical and viscoelastic control. Since the term was coined in the late 19th century, hydrogels have been widely used in drug ...
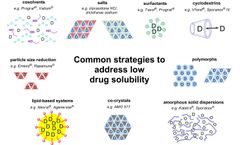
In the process of preparing drugs into liquid preparations, a common problem is that most drugs are poorly soluble in water. ...
The reason value-based agreements have created buzz is because gene, cellular and immunotherapies are risky endeavors for drug developers. To enable research, development and access to these high-priced drug therapies, biopharmaceutical companies understand that the way they do reimbursements must change. ...
While improved process may help, there are still problems related to the products themselves, such as the need to move from a population-based model of product development, in which one size fits all, to a more targeted approach based on specialty drugs for patient subpopulations. The old paradigm dominated by big pharma and blockbusters must evolve, in order for a new ...