Vaccine Quality Articles & Analysis
17 articles found
In the field of vaccine development, peptide vaccines, as an emerging vaccine platform, have gradually become the focus of research due to their advantages such as high safety, strong targeting, and customizability. ...
Vaccines have been essential in disease prevention, and the requirements for vaccine quality continue to increase. The application of advanced separation and purification technologies during the preparation of traditional and new vaccines is an effective means to improve vaccine efficacy and reduce side effects. ...
The advent of mRNA vaccines has revolutionized the field of immunization, offering a rapid and adaptable response to emerging infectious diseases. ...
The proportion of mRNA with cap structure can affect the immunogenicity and translation efficiency of mRNA, so the capping rate is a key quality indicator of mRNA vaccines or drugs.The molecular weight of mRNA is generally hundreds to thousands of KDa, while the cap structure is only about 500 Da. ...
Vaccines have been essential in disease prevention, and the requirements for vaccine quality continue to increase. The application of advanced separation and purification technologies during the preparation of traditional and new vaccines is an effective means to improve vaccine efficacy and reduce side effects. ...
However, despite the fact that aluminum adjuvants in vaccines have been used in enhancing vaccine immune responses for nearly decades, the molecular mechanisms of aluminum adjuvants are still not fully understood. ...
By understanding the multifaceted applications of glycerin in vaccine formulation, storage, and delivery, pharmaceutical manufacturers can optimize their vaccine production processes to ensure superior quality and effectiveness. ...
Raw enzymes orchestrate these modifications with precision, ensuring that the mRNA is well-prepared for its crucial role in vaccine production. Quality Assurance: The road to a successful mRNA vaccine is paved with quality control measures. ...
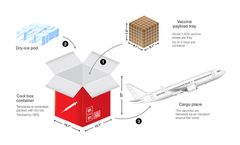
Due to the particularity of vaccines, vaccines need to be kept in a constant and suitable temperature environment during transportation and storage, and different vaccines require different environmental temperatures. Therefore, vaccines often need to be processed in the cold chain, and Temperature Humidity Recorders with ...
From the point of manufacturing until the last mile, the vaccine cold chain includes a series of interconnected steps: Manufacturing and quality control Vaccine manufacturers adhere to stringent quality control measures to ensure the vaccines’ potency and safety. ...
About the author Collected by CD BioGlyco, a biotechnology company dedicated to the development, preparation and quality control of polysaccharide vaccines and polysaccharide conjugate vaccines. Production of polysaccharide vaccines Produce polysaccharide vaccines based on the immunogenicity of ...
There are several potential COVID-19 vaccines that may soon be available for widespread distribution. In particular, the United Kingdom has recently approved Pfizer’s vaccine, and the U.S. ...
When shipped elsewhere, transportation is provided by a cold chain process in place to safeguard the quality of the medical products. Typically, cold chain operations employ of refrigeration units to maintain ideal conditions for vaccines or other life science products. Vaccine Cold Chain The purpose of the vaccine cold chain ...
ByAKCP
All vaccines are sensitive to extreme heat and extreme cold and require both proper vaccine storage and management to avoid any loss of potency. Immunization is the most effective and economical public health measure against infectious diseases that cause morbidity or mortality. Immunization prevents two to three million deaths every year from diseases such as hepatitis, pertussis (whooping ...
ByAKCP
Each vaccine needs to be stored in a cool and refrigerated place to ensure its efficiency and effectiveness are maintained during use. Every year, millions of people are vaccinated to strengthen their immunity and protect themselves from infectious diseases. According to the regulations of the World Health Organization (WHO), vaccination is an approved tool for dealing with deadly contagious ...
ByAKCP
” Bharat Biotech has commercialized 16 vaccines, including a vaccine developed against the H1N1 flu that caused the 2009 pandemic. FluGen will transfer its existing manufacturing processes to Bharat Biotech to enable the company to scale up production and produce the vaccine for clinical trials. Bharat Biotech will manufacture the ...
Data quality is widely considered as a very serious problem for the majority of companies due to the specificities of each business context and the lack of adapted solutions. ...