Vaccine Safety Articles & Analysis
26 articles found
In the field of vaccine development, peptide vaccines, as an emerging vaccine platform, have gradually become the focus of research due to their advantages such as high safety, strong targeting, and customizability. ...
Once synthesized, these proteins spontaneously assemble into VLPs, providing a scalable and efficient way to produce vaccines. Applications in Vaccination VLPs have shown considerable promise in the field of vaccination. ...
It allows for the identification of suitable donors and improved matching processes, thereby enhancing graft survival rates. Implications for Vaccine Development In vaccine development, concerns about cross-reactivity can influence the safety and effectiveness of the vaccine. Researchers aim to create vaccines ...
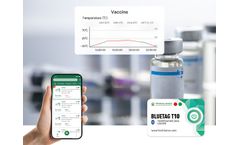
During the cold chain transportation of vaccines, serums, and biological products, 2-8℃ is a very suitable temperature. The vaccine Bluetooth temperature sensor provided by Freshliance has high accuracy. ...
Advantages of Recombinant Vaccines Recombinant vaccines come with several notable benefits: Safety: Since these vaccines do not contain live pathogens, there is a significantly reduced risk of causing disease in vaccinated individuals. ...
Lessons learned from COVID-19 treatments are being applied to other viral diseases, helping to develop more effective immune-based therapies. Vaccine Innovations Vaccine development has taken center stage in the fight against viral infections. ...
Vaccines have been essential in disease prevention, and the requirements for vaccine quality continue to increase. The application of advanced separation and purification technologies during the preparation of traditional and new vaccines is an effective means to improve vaccine efficacy and reduce side effects. Here are some commonly used purification methods: Centrifugal 1. ...
The advent of mRNA vaccines has revolutionized the field of immunization, offering a rapid and adaptable response to emerging infectious diseases. ...
Alfa Cytology has announced pancreatic cancer vaccine development services to improve the efficacy and safety of related vaccines. ...
To counter the threat of strains such as cVDPV, the new novel oral polio vaccine type 2 (nOPV2) was developed. It is designed to be genetically stable, therefore reducing the risk of reversion to a more virulent form and thereby improving overall safety and effectiveness. After rigorous testing and safety assessments, the vaccine ...
At present, the most effective vaccine is the mRNA vaccine developed by Pfizer-BioNTech and Moderna. ...
However, it has been demonstrated that sedimentation behavior severely affects the dispersion state of the mixture, which further affects the safety and stability of the vaccine. Granularity The two main factors controlling the performance of adjuvant suspensions are particle size and the charge of the dispersed particles. ...
Besides the advantages in terms of specificity and safety, peptide vaccines offer clear cost and supply advantages by reducing production costs and delivery times.However, the development of vaccines based on these minimal sequences is not without challenges. ...
This article explores the intricate and indispensable role that raw enzymes play in the manufacturing of mRNA vaccines, delving deep into their significance and impact on vaccine production. ...
From the point of manufacturing until the last mile, the vaccine cold chain includes a series of interconnected steps: Manufacturing and quality control Vaccine manufacturers adhere to stringent quality control measures to ensure the vaccines’ potency and safety. ...
Vaccine excipients play a crucial role in the development and manufacturing of vaccines. These are substances added to the vaccine formulation to enhance its stability, efficacy, and safety. Vaccine excipients can include adjuvants, stabilizers, preservatives, and additives. ...
Malaria vaccines have been under development since the 1960s and have faced many obstacles: the malarial parasites produce very complex antigens which made developing a vaccine very challenging. ...
[ii] Ensuring flu vaccines remain effective through proper storage not only helps guarantee that those who are vaccinated are protected but also makes people more likely to continue opting to receive vaccines. ...
Subjects will be randomized 2:1 to receive a single dose of M2SR or placebo, delivered intranasally, and all subjects will receive a single dose of inactivated quadrivalent influenza vaccine (QIV) at least 28 days later. The primary endpoint is to measure the safety and tolerability of M2SR through 6 months post-vaccination. ...
All vaccines are temperature sensitive and require the proper environmental conditions to ensure vaccine stability. Exposure to extreme heat or cold can be catastrophic to millions of vulnerable people, especially children, who would otherwise go unvaccinated. To ensure the safety and potency of vaccines, the VFC Program, which ...