Vaccine Storage Articles & Analysis
24 articles found
We built a more than 700,000-cubic-foot vaccine storage solution for a global manufacturer, during COVID-19 lockdown, on time and on budget. ...
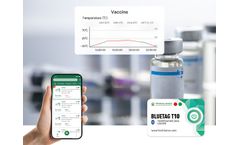
The optimal storage environment temperature for most vaccines is 2-8℃. It is necessary not only to control the temperature stability but also to monitor the temperature. The 2-8℃ Vaccine Bluetooth Temperature Sensor can help managers view the storage environment temperature data directly on the APP without opening the incubator, ...
By understanding the multifaceted applications of glycerin in vaccine formulation, storage, and delivery, pharmaceutical manufacturers can optimize their vaccine production processes to ensure superior quality and effectiveness. ...
Our solutions are not mere tools, but rather lifelines that ensure the safe storage and transport of critical drugs, vaccines, samples and more. The efficacy of these biologicals, crucial in medical treatments and procedures, is inextricably linked to the quality and reliability of the storage and transport solutions used along their supply ...
Due to the particularity of vaccines, vaccines need to be kept in a constant and suitable temperature environment during transportation and storage, and different vaccines require different environmental temperatures. ...
Maintaining proper temperature conditions during production is critical to preserving the vaccine’s integrity. Storage and transportation After production, vaccines are stored in temperature-controlled medical devices, such as refrigerators and freezers, to maintain their stability. ...
Malaria vaccines have been under development since the 1960s and have faced many obstacles: the malarial parasites produce very complex antigens which made developing a vaccine very challenging. ...
Vaccine Freezers are required for the safe storage of vaccines that must be kept at extremely low temperatures. Without sufficient storage, a vaccine is likely to form deviations, drastically impacting the vaccines efficiency. ...
COVID -19 Vaccine Overview The development of a COVID-19 vaccine by drug manufacturers has been welcomed across the globe. ...
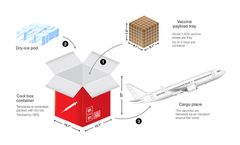
Transportation Specifically, let’s look at the COVID-19 vaccine storage and handling file to see what they are required to know.ll of the ult and freezer storage units that will be storing the COVID-19 vaccine must be set up to stabilize temperatures at the recommended temperature range specified by the manufacturer before ...
ByUbiBot
[ii] Ensuring flu vaccines remain effective through proper storage not only helps guarantee that those who are vaccinated are protected but also makes people more likely to continue opting to receive vaccines. ...
Not only does this leave them unprotected, it also degrades the trust in vaccinations. When it comes to the last link in the cold chain, proper vaccine storage is only achieved by using a quality pharmaceutical refrigerator. B Medical Systems Pharmacy Refrigerator and how they can support vaccine acceptance B Medical ...
The new TCW80SDD Vaccine Refrigerator will allow to keep vital vaccines refrigerated in places without electrical infrastructure, making vaccinations more equitable around the world. ...
To control and prevent this virus and others of its kind, vaccination has been adopted as an effective way to protect public health. Vaccines are chemical and biological products, which means they are sensitive to both cold and heat and must be kept in cold storage throughout the vast cold chain. The vaccine cold chain is ...
All vaccines are temperature sensitive and require the proper environmental conditions to ensure vaccine stability. ...
For research facilities, hospitals, clinics, doctor offices and other medical establishments, vaccine storage is absolutely critical to ensuring the accuracy and efficiency of vaccines administered. Although regulatory organizations carry out standard audits to examine vaccine storage procedures, it is important ...
The vaccine cold chain begins with a cold storage unit at the vaccine manufacturing plant, extends to the transport and delivery of the vaccine (including proper storage at the provider facility), and ends with the administration of the vaccine to the patient. ...
In this particular period of the year the company works close to the manufacturers of freezers to help them in providing high quality units for the storage of Covid-19 vaccine. Vaccines require unbroken and accurate refrigeration systems to remain efficacy and safe and the real challenge is to ensure reliability in the long term. ...
Unlike medicines, which are used to treat or cure diseases, vaccines are intended to prevent them. Handling and Storage of Vaccines Developing a vaccine can take years before it is deemed safe for human use and, thereafter manufactured and made available for widespread distribution and inoculation. ...
On November 9, 2020, Pfizer and BioNTech announced that their trial of a Covid-19 vaccine showed a 90% efficacy rate and very promising results in participants who had developed symptoms. ...