Refine by
Allograft Suppliers & Manufacturers
79 companies found

based inParsippany, NEW JERSEY (USA)
Based in Parsippany N.J., Onkos Surgical is a privately held, specialty medtech company founded in 2015. We believe that individuals with cancer requiring surgery deserve solutions designed specifically for them. This principle is the driving force ...
The scaffold is comprised of a proprietary blend of microparticulate cortical, cancellous and demineralized cortical allograft bone. Optimal particle sizes range from 100-300 microns. Via Graft processing preserves a cell population ...

based inSeattle, WASHINGTON (USA)
CorneaGen is a mission-driven company committed to transforming how corneal surgeons treat and care for the cornea. Based in Seattle, the company is innovating the next generation of cornea care, from new medical devices and biologics to ...
The all-new EndoSerter-PL® is preloaded with tissue processed to the surgeon’s exact specifications with saves precious time in the O.R. This product is an innovation for the next generation of DSEK ...

based inSouth San Francisco, CALIFORNIA (USA)
CareDx: Transforming Transplant Patient Care Through Novel Surveillance Management Solutions. Our understanding of transplant patients and care teams allows us to develop solutions to extend graft life. With two decades of commitment to transplant ...
KidneyCare is a multimodality surveillance solution for kidney transplant patients. It includes AlloSure® Kidney, AlloMap Kidney, and KidneyCare ...

based inLund, SWEDEN
BONESUPPORT AB is a Scandinavian orthobiologics company that develops and markets CERAMENT®, an innovative range of radiopaque injectable osteoconductive and drug-eluting bioceramic products that have a proven ability to heal defects by remodeling ...

based inClearwater, FLORIDA (USA)
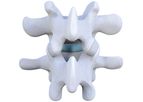
Zentech Spine is a medical device company located in Clearwater, Florida. Zentech's Zenfuze Posterolateral Lumbar Fusion Procedure is a minimally invasive surgical solution that shows the ability to reorient and stabilize the spine, promote fusion ...
The Zenfuze System is a minimally invasive solution for patients suffering from low back pain. Following conservative treatment methods, Zenfuze may be an option. The Zenfuze System can treat conditions such as spinal stenosis, spinal instability, ...

based inWestminster, COLORADO (USA)
Cerapedics is dedicated to enhancing the science of bone repair by staying at the leading edge of osteobiologic science and product development. Our lead technology platform (i-FACTOR Peptide Enhanced Bone Graft) is the only biologic bone graft that ...

based inBurlington, MASSACHUSETTS (USA)
LeMaitre is a leading global innovator, manufacturer and marketer of devices for the treatment of peripheral vascular disease. We’re proud to provide vascular surgeons with the solutions they need to diagnose and heal their patients. While LeMaitre ...
RestoreFlow Allografts provide a high quality allograft tissue using state-of-the-art technologies for tissue restoration and preparation techniques. These allografts can be used for cardiac repair ...

based inWest Chester, PENNSYLVANIA (USA)
Centinel Spine develops, tests and markets new and innovative products with an eye toward making spinal fusion surgery easier for both surgeon and patient alike. Centinel Spine derived its name from the “Sentinel Sign” — the radiographic ...
3D-printed porous titanium cervical interbody with 2-level clearance! lexible: FLX implants include a solid exoskeleton for added strength, along with a proprietary lattice scaffold designed to have a stiffness similar to bone. Reduced device ...

based inCorona, CALIFORNIA (USA)
For over 20 years, United Endoscopy has been a leader in providing state-of-the-art refurbished medical equipment and advanced equipment repair to its customers worldwide. Since its founding in 1996 by Jeremy Cooper, United Endoscopy has ...

based inYenimahalle/ANKARA, TURKEY
BIOLOT Medical is a company based on pain management, tissue regeneration, bone regeneration and wound healing. We aim to provide innovative medical solutions that make patients look younger, feel more active and remain active. It is our quality and ...
Hyalocover is an absorbable mesh of fabric. It can be used in orthopedic trauma and reconstruction, soft tissue regeneration in surgical treatment and tendon ...

based inBedford, MASSACHUSETTS (USA)
Anika is a global joint preservation company that creates and delivers meaningful advancements in early intervention orthopedic care. We partner with clinicians to understand what they need most to treat their patients and we develop minimally ...
Anika’s SpeedSprial® CMC Allograft System utilizes a pre-shaped, dense, strong, and flexible allograft implant to treat thumb CMC joint pain and/or instability caused by osteoarthritis (OA). The ...

based inMarietta, GEORGIA (US) (USA)
MiMedx is a biopharmaceutical company developing, manufacturing and marketing biologics utilizing human placental-based allografts for multiple sectors of healthcare. “Treating more patients with better outcomes” is the framework behind our mission ...
For patients with challenging wounds (e.g. non-healing ulcers) or requiring surgical procedures, placental-based therapies are a valuable treatment option. Clinical evidence from many randomized clinical trials have demonstrated that placental-based ...

based inMiami, FLORIDA (USA)
We are the regenerative medicine company that develops and delivers innovative allograft solutions designed to support the body’s healing and regenerative potential to help improve patient outcomes and quality of life. VIVEX Biologics processes, ...
A flexible, multi-layer allograft maintaining the amnion, its intermediate/spongy layer and the chorion layers of the amniotic sac, providing improved handling and increased workability. ...

based inMarietta, GEORGIA (US) (USA)
MIMEDX is an industry leader in advanced wound care and a therapeutic biologics company. As a company, we are driven to bring hope to people with hard-to-heal wounds. Our ability to advance science will further differentiate the value of our ...
AMNIOCORD provides a protective environment to support the healing process. AMNIOCORD is derived of human umbilical cord. It is a thick graft that allows for suturing to keep the graft in place and works with deep surgical sites. ...

based inStillwater, MINNESOTA (USA)
Vascudyne is developing, manufacturing and commercializing regenerative acellular in-vitro grown “allograft” tissue. We are visionaries and realists in the right proportions. We are after the holy grail of bringing tissue-engineered medical products ...
Our proprietary TRUE Tissue technology platform is currently available for investigational use only. TRUE Tissue developed from cells isolated from donor tissue and is 100% biological. There are no synthetic materials or chemical fixation used, and ...

based inOakville, ONTARIO (CANADA)
We bring innovation and vision to practice globally through principled people. For over 60 years we’ve been world-wide leaders providing advanced eye care products, solutions, and resources for eye care professionals and their patients. Labtician ...
LabAmnioTM is processed, dehydrated, sterilized human amniotic membrane allograft intended for use in ocular tissue repair. ...

based inPinebluff, ALABAMA (USA)
ATEX Technologies specializes in manufacturing advanced textile components for implantable medical devices. Their expertise lies in blending state-of-the-art textile manufacturing with precise engineering to meet stringent medical industry ...
Their primary utility lies in structural stabilization and reinforcing weak or damaged tissues post-major repairs, such as in allograft placements or fracture repairs. Additionally, these versatile cables serve as effective ...

based inSeoul, SOUTH KOREA
CG Bio Inc. is a total healthcare provider with a goal of enhancing the quality of life by offering range of innovative solutions for the spectrum of spinal disorders and wound management. Through extensive research activity, CG Bio is a leading ...

based inMemphis, TENNESSEE (USA)
In 2014, three tenured product executives founded CrossRoads Extremity Systems to launch breakthrough products for faster healing. They wanted to create a company based on their values and what they enjoyed most about being medical device engineers. ...
The CrossTie™ Intraosseous Fixation System is indicated to aid in the fixation of fractures, fusions, and osteotomies of the toes, such as hammertoe, claw toe, mallet toe and inter-digital fusions. There are potential risks ...

based inRichardson, TEXAS (USA)
Fuse Medical is a manufacturer and national distributor of medical devices, providing a broad portfolio of orthopedic implants including: internal and external fixation products; upper and lower extremity plating and total joint reconstruction; soft ...