- Home
- Companies
- usa arkansas
- catheter delivery
Show results for
Refine by
Catheter Delivery Suppliers Serving Arkansas
50 companies found

based inShatin, N.T., CHINA
We are a global market leader in the development and manufacture of PCI/PTA balloons. Headquartered in Hong Kong, China, we sell products to over 70 countries and regions worldwide. In addition to PCI/PTA balloons, we also specialize in coronary ...
The Pro-Healing DES Designed for rapid vessel healing and superior clinical ...

based inOakville, ONTARIO (CANADA)
CHS Ltd. (est. 1967) is a privately held medical device manufacturer and specialty distributor located in Oakville, Ontario. Customers served are in the acute hospital and non-acute healthcare space in Canada and Internationally. CHS ...

based inHuntington, NEW YORK (USA)
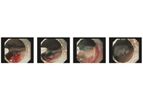
Thoracent specializes in providing pulmonary and thoracic products to the U.S. medical market. The BONASTENT tracheobronchial stent is our flagship product. The BONASTENT stent features a nitinol woven design that is fully covered with an ultra-slim ...
The BONASTENT tracheobronchial stent incorporates the patented “hook & cross” nitinol stent design with a silicone cover purposely placed across the entire interior of the stent to prevent in-growth. The stent is delivered with an ...

based inAbbott Park, ILLINOIS (USA)
We create breakthrough products – in diagnostics, medical devices, nutrition and branded generic pharmaceuticals – that help you, your family and your community lead healthier lives, full of unlimited possibilities. Today, 109,000 of us are working ...

based inMontreal, QUEBEC (CANADA)
QXMedical was founded in 2007 in Montreal, Canada on the principle of providing uncompromised value and quality in underserved high-growth segments of the medical device market. Today, the company is known for its innovative solutions for complex, ...

based inSanta Clara, CALIFORNIA (USA)
EndoClot Plus, Inc. (EPI) is a privately held medical device company in Santa Clara, California. In collaboration with endoscopists and biomaterial scientists, EPI developed EndoClot Polysaccharide Hemostatic System (EndoClot PHS), a breakthrough ...
EndoClot PHS is a single use medical device consisting of Absorbable Modified Polymers (AMP) and a unique powder delivery system (applicator). It is a safe and effective method to control bleeding in both upper and lower GI tracts. It works ...

based inMinneapolis, MINNESOTA (USA)
At Medtronic, we believe in the power of medical technology to improve lives. Seven decades ago, our co-founder invented the battery-powered pacemaker. Today, we are among the largest medical device companies in the world. With operations in 150 ...
The Marathon™ flow directed microcatheter was designed for the infusion of therapeutic agents such as embolization materials and contrast media in tortuous, distal vessels.1 . The ApolloTM OnyxTM delivery microcatheter was expressly designed ...

based inMelville, NEW YORK (USA)
FONAR was incorporated in 1978, making it the first, oldest and most experienced MR manufacturer in the industry. FONAR introduced the world's first commercial MRI (a whole-body MRI scanner) in 1980, and went public in 1981. In 1982, FONAR ...

based inHorsham, UNITED KINGDOM
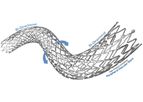
Veryan has developed innovative technology to improve the performance of vascular stents by adopting the principle of biomimicry; developing structures that imitate those occurring naturally. Veryan’s vascular biomimetic stent technology involves ...
The Challenge: Peripheral Arterial Disease. People are living longer and with an increase in the incidence of obesity, diabetes and other contributing factors, claudication may cause substantial tissue changes and, in some scenarios, can lead to ...

based inWinsen, GERMANY
QualiMed was found in 1997 as an OEM manufacturer for implantable medical devices with a focus on the development and regulatory approval of coronary stents and their respective delivery devices. Later the business was expanded to peripheral ...
The NAVALISADVANCE Peripheral Vascular Self-Expanding Stent System combines highly durable nitinol stent with a flexible delivery system to provide proven clinical ...

based inRedwood City, CALIFORNIA (USA)
A global leader in interventional pulmonology, planning tools, and treatments for obstructive lung disease. Based in Redwood City, California, and Neuchâtel, Switzerland, Pulmonx is the maker of the Zephyr Endobronchial Valve. The Zephyr® Valve was ...
Sizing and Placement in One Step: Easy to use. Quick and accurate sizing of the length and width of the airways. Equivalent catheter sizes to valve sizes. Available in a “J” configuration to allow the bronchoscope access to more ...
based inMarlborough, MASSACHUSETTS (USA)
Boston Scientific is dedicated to transforming lives through innovative medical solutions that improve the health of patients around the world. Our work is guided by core values that define Boston Scientific culture and empower our employees. Built ...
We took your feedback and designed the new ZURPAZ 8.5F Steerable Sheath to not only meet, but exceed, your expectations for catheter ...

based inSan Mateo, CALIFORNIA (USA)
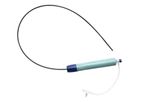
Founded in 2015, Route 92 Medical is a privately held, commercial-stage medical technology company, based in San Mateo, CA. We are developing novel neuro-interventional technology with an application in stroke. Route 92 Medical’s mission is to ...
The Route 92 MedicalTenzing 7 Delivery Catheter is a single-lumen, variable stiffness catheter with a long, tapered tip delineated by two radiopaque markers. The Delivery ...

based inKennesaw, GEORGIA (US) (USA)
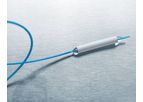
Our purpose is to develop simple, elegant solutions that address cardiac and vascular surgeons’ most difficult challenges in treating patients with diseases of the aorta and to deliver breakthrough technologies of unsurpassed quality that have ...
Thanks to the use of cobalt chromium, the E-ventus BX is suitable for follow-up MRI examinations. The E-ventus BX delivery system has a highly flexible catheter which allows easy advancement in the vessel and ...

based inOaks, PENNSYLVANIA (USA)
VascuTech Medical, LLC has evolved from a consulting firm serving major customers such as C.R. Bard, to a contract manufacturer of FDA and ISO 13485 registered medical device manufacturer serving OEMs and healthcare providers in interventional ...
Intravascular Drug Delivery Catheters are intended for the local delivery of various therapeutic and diagnostic agents into the coronary and peripheral vasculature. VascuTech Medical is a leading ...

based inAlpharetta, GEORGIA (US) (USA)
Spectrum is your first-call solutions provider for development through scaled manufacturing of critical polymer-based components and devices for medical and other demanding markets. We design solutions that support life-saving procedures and satisfy ...
With hundreds of materials and tools in stock and countless configurations readily available, we can rapidly produce prototypes to accelerate your extrusion, catheters, additive manufacturing, and 3D medical printing development ...

based inReutlingen, GERMANY
It is probably a combination of many factors that has made us successful since 2004: the good collaboration of a medium-sized family company; the site in the heart of Germany’s medical technology region around the University of Tübingen; our ...
The semi-flexible hybrid cell Cobalt Chromium bare stent has been specifically designed for the effective treatment of Coarctation. Available in XL and ...
based inZhuhai, CHINA
NeuroSafe Medical Co., Ltd. specializes in the development of state-of-the-art medical technologies designed to improve patient outcomes and advance modern healthcare solutions. With a commitment to ensuring safety and efficacy, the company focuses ...
This injectable radiopaque embolic agent comprises EVOH (ethylene vinyl-alcohol copolymer), DMSO (dimethyl-sulfoxide), and micronized tantalum powder, facilitating exceptional visibility under fluoroscopy for precise delivery. The ...

based inLake Forest, CALIFORNIA (USA)
NaviGate Cardiac Structures, Inc., (NCSI) is an early-stage company focused on developing transcatheter solutions for the treatment of atrioventricular valve regurgitation. The NaviGate valve-based technology was licensed from Cleveland Clinic and ...
The GATE™ System is intended to treat functional tricuspid regurgitation (FTR)/tricuspid insufficiency by replacing the function of a patient’s dysfunctional native tricuspid valve with a bio-prosthetic atrioventricular valved stent ...

based inAdelaide, AUSTRALIA
Established in 2016 out of a desire to pursue optimal medtech solutions and to challenge the status quo, Numedico is a globally focused safety medical devices manufacturer and distributor based in Australia. We provide high quality medical supplies ...
This high quality retractable syringe is like no other on the market, and is available at a fraction of the price. ClickZip technology prevents reuse and needlestick injury due to our innovative titled needle technology that encapsulates the needle ...