- Home
- Companies
- canada yukon
- in vitro diagnostic use
Refine by
In Vitro Diagnostic Use Suppliers Serving Yukon
50 companies found

based inBoston, MASSACHUSETTS (USA)
At PathAI, we’re dedicated to improving patient outcomes with reliable AI-Powered technology and meaningful collaboration with biopharma, laboratories, and clinicians — aiming to provide patients with access to accurate diagnoses and effective ...
AISightTM is a web-based digital pathology slide viewing platform that enables the management and analysis of whole slide ...

based inWenzhou, CHINA
Global player specialized in manufacture and distributor of medical products, with a presence in 36 countries. A group with five (5) subsidiary factories and eight (8) joint venture manufacture base. A product scope more than 110 products, with a ...

based inWindsor, NOVA SCOTIA (CANADA)
BioMedica Diagnostics is a specialized company offering diagnostic solutions with a strong focus on Thrombosis and Haemostasis. The company collaborates globally with technology development firms and healthcare stakeholders to engineer and ...

based inWaltham, MASSACHUSETTS (USA)
Nova Biomedical is a company dedicated to using advanced technology to develop better blood testing analyzers for medical use. These products are called in vitro diagnostic devices (IVD). We use some of the same advanced technology to provide the ...
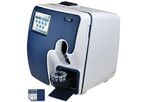
Exceptional Simplicity in a New Generation Electrolyte Analyzer. Introducing a new generation electrolyte analyzer that combines the revolutionary microelectronics of the consumer world with new microsensor technology for a simpler, smaller, faster, ...

based inMumbai, INDIA
Tosoh India Pvt. Ltd., based in Mumbai, the commercial capital of India, is a subsidiary of Tosoh Corporation. Headquartered in Tokyo, Japan, Tosoh Corporation is a conglomerate of diversified world class companies operating across the globe. ...

based inSALUGGIA, ITALY
We are global leader in the laboratory diagnostics market, specializing in the immunodiagnostics and molecular diagnostics segments. For over 50 years we have been dealing with diagnostics to help physicians and patients make better decisions in the ...
Focus Diagnostics’ Simplexa™ BKV Molecular Control consists of a DNA fragment to a well conserved region of BKV VP2 gene intended to be used as a control in molecular testing. This control is not intended for use as a control with ...

based inUdine, ITALY
Ulisse BioMed is an Italian healthcare biotech company with corporate offices in Udine, Italy and with R&D and Manufacturing labs in Trieste, Italy. Ulisse BioMed is creating novel, innovative and transformative diagnostic platforms and products. ...
The assay is intended for the qualitative detection of RNA from SARS-CoV-2 in clinician-collected nasopharyngeal swab specimens from patients with signs and symptoms suggestive of COVID-19 (e.g., fever and/or symptoms of acute respiratory ...

based inSan Diego, CA 92126 USA, CALIFORNIA (USA)
America Diagnostics, Inc. is a California company that develops and produces IVD devices and diagnostics raw materials. The quality of AmeriDx products is guaranteed by its quality management system. All the products are manufactured in a clean ...
This product is for in vitro diagnostic use, following guidance from the FDA for Emergency Use Authorizations of tests submitted for approval on March 16, 2020. This test has not ...

based inEpalinges, SWITZERLAND
At Abionic we are focused on delivering true patient-centric diagnosis solutions, which means a rapid, reliable test available at any time, from anywhere, whenever a patient needs it… A potential to save millions of lives is what drives us to work ...

based inKAARINA, FINLAND
Labmaster Oy Ltd. is a company engaged in the design, development, and manufacturing of various products, ensuring they meet specified criteria through proper storage, usage, and transportation guidelines. They emphasize adherence to instructions ...
The Labmaster LUCIA™ MxA Kit is a sophisticated in vitro diagnostic tool used for the quantitative assessment of the Myxovirus resistance protein A (MxA) in whole blood samples. This ...

based inNew York City, NEW YORK (USA)
Led by a team of experts in the fields of life sciences, oncology, pathology, technology, machine learning, and healthcare, Paige strives to transform cancer diagnostics. We make it possible not only to provide additional information from digital ...

based inSeattle, WASHINGTON (USA)
InBios is a leading biotechnology company based in Seattle, Washington (USA). At InBios, we believe an accurate diagnosis is the first step to effective treatment. Specializing in the design, development and manufacture of immunodiagnostic devices ...
The Chagas DetectTM Plus (CDP) Rapid Test is a rapid immunochromatographic strip assay for the qualitative detection of human lgG antibodies to Trypanosoma cruzi (T. cruzi) in human serum and whole blood matrices (venous and capillary (finger prick) ...

based inLake Forest Park, WASHINGTON (USA)
IEH was founded in 2001 by Dalia Alfi and Mansour Samadpour, who left their positions at the University of Washington, School of Public Health, to start the company. It is a family-owned and operated company and has grown rapidly since 2001, both ...

based inCoralville, IOWA (USA)
A champion of life sciences research for more than 30 years, IDT develops and manufactures nucleic acid products that support the life sciences industry. Areas of focus include academic and commercial research, agriculture, medical diagnostics, ...
We have combined our proven oligo manufacturing expertise and ISO 13485 certified production processes to deliver GMP-manufactured MGB Eclipse Probes and companion primers. Together, these probe and primer pairs are ideal for genotyping qPCR assays. ...

based inPoway, CALIFORNIA (USA)
Diazyme uses its proprietary enzyme technologies to develop diagnostic reagents which can be used on most automated chemistry analyzers in user-friendly formats. Diazyme's products include test kits for diagnosis of cardiovascular disease, liver ...

based inXiamen, CHINA
Amoy Diagnostics Co., Ltd. (AmoyDx) is an R&D based diagnostic company, focusing on molecular diagnostics for oncology precision medicine. With completely independent intellectual property rights of ADx-ARMS®, Super-ARMS®, ddCapture® and ADx-HANDLE® ...
Lung cancer is one of the most common malignant tumor, and 80~85% of lung cancers are non-small cell lung cancer (NSCLC). There are many driver mutations in NSCLC. The frequency of mutations in NSCLC for EGFR, HER2, KRAS and BRAF genes are ...

based inSan Antonio, TEXAS (USA)
At Germaine Laboratories, we provide high-quality, reliable diagnostic testing supplies and services. We partner with our network of distributors to make sure we get the right product into the right hands at the right time. Browse our product ...
Ref #78200 For the quantitative determination of hemoglobin in non-anticoagulated capillary whole blood or anticoagulated venous whole blood. CLIA ...

based inVienna, AUSTRIA
ViennaLab Diagnostics specializes in easy-to-use in vitro diagnostic assays for the detection of genetic variants associated with genetic disorders, genetic predispositions, pharmacogenetics, cancer, and the human microbiome. Our focus is on product ...
A reliable tool to personalize therapy of lung cancer patients with acquired TKI-resistance mutation T790M in the EGFR ...

based inPaoli, NEW JERSEY (USA)
Beta Lifescience is a professional biotech company committed to providing premium quality life science research reagents worldwide across research and diagnostic markets. Our strong portfolio of recombinant proteins, viral antigens, antibodies, ...
The Anti SARS-COV2 / 2019-nCoV NP mAb (IgG&IgM Positive Control) product contains two isotype antibody clone Human IgG and IgM, which can recognizes2019-nCoV Nucleocapsid ...

based inNanjing, CHINA
Founded in 2002, Jiji Biotech Co., Ltd.is a medical and health industry chain that integrates independent research and development, large-scale production and professional marketing. It has many subsidiaries and offices at home and abroad. the ...
Can be used as a aid in identifying individuals with an adaptive immune response to SARS-CoV-2, indicating recent or prior infection, it should not be used to diagnose acute SARS-CoV-2 infection. ...