Refine by
Transcatheter Valve Suppliers & Manufacturers
30 companies found

based inAachen, GERMANY
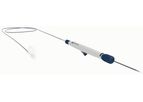
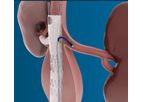
At Protembis, we strive to develop a simple and reliable solution to protect patients from brain injury during left-sided heart procedures. With our product, the ProtEmbo® Cerebral Protection System – already in its second generation – it is our ...
ProtEmbo is a catheter-based cerebral protection system to deflect embolic material away from the cerebrovascular circulation during leftsided heart procedures – in particular transcatheter aortic valve ...

based inStillwater, MINNESOTA (USA)
Vascudyne is developing, manufacturing and commercializing regenerative acellular in-vitro grown “allograft” tissue. We are visionaries and realists in the right proportions. We are after the holy grail of bringing tissue-engineered medical products ...
Bioprosthetic heart valves may have addressed some of the limitations of mechanical valves but still carry their own: gradual degeneration such as pannus growth, leaflet fibrosis and calcification, delamination of the connective tissue, and ...

based inToowong, AUSTRALIA
Anteris is a structural heart company delivering clinically superior solutions that help healthcare professionals create life-changing outcomes for patients. Our focus is on investing in and developing next-generation technologies with world class ...
The World’s First & Only 3D Single-piece Aortic Valve. DurAVR™ heart valve has the potential to deliver a functional cure in the treatment of Severe Aortic Stenosis. The unique design of DurAVR™ coupled with the ADAPT® ...

based inSanta Clara, ALABAMA (USA)
Abbott is dedicated to advancing structural heart therapies in collaboration with globally recognized specialists and care teams. The company's focus is on repairing the effects of structural heart diseases through innovative therapies that ...
The Epic™ Max Aortic Stented Tissue Valve represents the latest advancement in aortic valve replacement technology, engineered to optimize clinical outcomes and enhance lifetime patient management. It features ...

based inHighland Hts., OHIO (USA)
Since Norman Noble commenced business operations in 1946, we have been proud to manufacture and innovate medical implants and devices. Over 75 Years of Manufacturing Medical Implants and Devices From humble beginnings in Cleveland, we have since ...
Proprietary laser machining technologies are custom built by Norman ...

based inSingapore, SINGAPORE
For more than 25 years, the Biosensors International Group (“Biosensors”) has been developing and marketing critical care catheter systems and related devices used during heart surgery and intensive care treatment. Continually striving to be at the ...
The IMPERIA Delivery System, a solution by NVT GmbH, is specifically engineered for the efficient transfemoral implantation of the ALLEGRA Transcatheter Heart Valve. This advanced system is distinguished by its ...

based inJiaxing City, CHINA
We, Best Hope Precision Device Co., Ltd. are engaged in design and manufacture precision injection molds, typically with rich experience for medical devices. A Leading Manufacturer of Molds and Injection Molded Parts for Medical Devices. In past ten ...
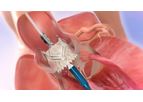
Aortic stenosis is a common disease where one of the heart valves narrows, restricting the flow of blood into the body. When it gets bad enough, the diseased valve often needs to be ...

based inSanta Clara, CALIFORNIA (USA)
Shockwave is focused on developing and commercializing products intended to transform the way calcified cardiovascular disease is treated. Shockwave aims to establish a new standard of care for the interventional treatment of atherosclerotic ...

based inMountain View, CALIFORNIA (USA)
Moray Medical is working to improve the quality of life for the millions suffering with leaky heart valves. Using our proprietary microfluidic robotics, more cardiologists can safely and cost-effectively treat the large number of patients who need ...
Moray Medical’s initial product is the Coral System, which includes a reusable Driver Assembly, a single-use Clipper toolset, and a Trident input device. The Clipper toolset includes a cardiac-valve-therapy edge-to-edge clip implant that is ...

based inKennesaw, GEORGIA (US) (USA)
Our purpose is to develop simple, elegant solutions that address cardiac and vascular surgeons’ most difficult challenges in treating patients with diseases of the aorta and to deliver breakthrough technologies of unsurpassed quality that have ...
CryoValve SG Pulmonary Human Heart Valves are indicated for the replacement of diseased, damaged, malformed, or malfunctioning native or prosthetic pulmonary valves. They may also be used in the replacement of native pulmonary valves when the Ross ...

based inIrvine, CALIFORNIA (USA)
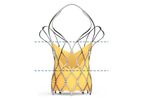
JenaValve is a clinical-stage medical device company developing the first transcatheter heart valve technology that is uniquely designed for the minimally invasive treatment of both severe aortic regurgitation and stenosis. JenaValve is passionate ...
Nitinol Support Frame: Self-Expanding Design – Available in (3) Sizes. Large Cells for Coronary ...

based inMississauga, ONTARIO (CANADA)
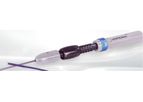
Baylis Medical Technologies is a company focused on developing and commercializing advanced medical devices used in endovascular therapy and other vascular procedures. Their products, such as the PowerWire Pro RF Guidewire, are designed to help ...
The PowerWire® Pro RF Guidewire is designed for precision and control in complex vascular procedures, aiding the introduction of diagnostic and therapeutic devices into various parts of the human vasculature, such as intracardiac, renal, and ...

based inIrvine, CALIFORNIA (USA)
Edwards Lifesciences is the global leader in the science of heart valves and hemodynamic monitoring. Driven by a passion to help patients, the company partners with clinicians to develop innovative technologies in the areas of structural heart ...
Designed for predictable deployment. The SAPIEN XT valve may be used in the aortic position via a broad range of access routes - transfemoral, transapical and ...

based inVienna, AUSTRIA
P+F Products + Features GmbH is a medical technology company focused on research, development, and manufacture of innovative minimally invasive solutions in the field of cardiology. Our R&D capabilities are the foundation of our approach to ...
The Vienna Aortic Self-Expandable Transcatheter Valve is designed to treat severe aortic stenosis in patients at medium to high risk for open heart surgery. The minimally invasive heart valve ...

based inMilano, ITALY
InnovHeart, with locations in Milan, Italy, and Boston, Massachusetts, develops transcatheter mitral valve replacement (TMVR) systems to treat patients suffering from mitral valve disease. The company is currently in clinical trials testing its ...
The Mitral Valve is a complex structure positioned between the left atrium and the left ventricle. Its function is to make unidirectional the blood flow from the left atrium to the left ventricle. The Mitral Valve can be affected by two major ...

based inLouisville, COLORADO (USA)
Colibri Heart Valve LLC (“Colibri”) is a privately held medical device company that researches and develops novel, patent protected, structural heart technologies. Colibri was formed in 2010 and is located near Boulder Colorado. Through Colibri’s ...
The Colibri Transcatheter Aortic Valve Implantation (“TAVI”) System is expected to redefine transcatheter aortic heart valve implantation as it is the ONLY, true low ...

based inSurat, INDIA
SMT is a global medical device company committed to making advance medical technologies accessible to everyone around the world. With a presence in over 69 countries, SMT has achieved recognition from the Government of India for its tremendous ...
Self-expanding TAVI device to have 2 rows of marker; First Markers are located at Node 1. Second Markers are located at Node 3. First marker helps; In precise implantation of the valve at the targeted implantation zone. To ascertain the depth of ...

based inLake Forest, CALIFORNIA (USA)
NaviGate Cardiac Structures, Inc., (NCSI) is an early-stage company focused on developing transcatheter solutions for the treatment of atrioventricular valve regurgitation. The NaviGate valve-based technology was licensed from Cleveland Clinic and ...
The GATE™ System is intended to treat functional tricuspid regurgitation (FTR)/tricuspid insufficiency by replacing the function of a patient’s dysfunctional native tricuspid valve with a bio-prosthetic atrioventricular valved stent ...

based inHouston, TEXAS (USA)
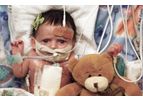
Since 2014, we have led the effort to improve children's lives with polymeric transcatheter valves (PTV) for children. PTVs are easy to manufacture in pediatric and adult sizes, expandable to accommodate growth of children, and reduces a need for ...
Congenital heart disease (CHD) is the most common birth defect and number one cause of infant mortality in the developed world. Twice as many children die from CHD than all childhood cancers combined. Our aim is to transform the care of children ...

based inBrunswick, AUSTRALIA
Avania is an integrated global, full-service CRO with specialized expertise in medical device, novel technology, and combination products. They advance products from feasibility all the way through post-approval trials in analytics, clinical trials, ...
Worldwide, heart disease is the leading cause of death. This is a primary reason why our commitment to your cardiovascular trial couldn’t be stronger. Whether your development program is geared toward treatment or prevention, you will have our ...