Refine by
Vascular Access Suppliers & Manufacturers
75 companies found

based inHeilbronn, GERMANY
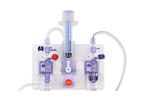
Xenios AG - a company of the Fresenius Medical Care Group - is a pioneer in extracorporeal heart and lung support. The Novalung and Medos products offer a wide range of heart and lung support products via a single platform, the Xenios console. ...
The iLA Membrane Ventilator is an extrapulmonary ventilation system which is used primarily to remove carbon dioxide. The heart pumps blood through it as it does through a natural ...

based inMiami, FLORIDA (USA)
At Gaumard, our mission is to empower healthcare providers with innovative training solutions to accelerate progress toward safer patient care and improved patient outcomes for everyone. We focus on developing state-of-the-art patient simulators, ...
Pediatric HAL® is the world's most advanced pediatric patient simulator and the first capable of simulating lifelike emotions through dynamic facial expressions, movement, and speech. HAL is designed to help providers of all levels develop the ...

based inNORTH MIAMI, FLORIDA (USA)
Located in South Florida, our headquarters in Miami city means something more than vacations or good time, our privilege location let us attend a vast community health care related over US and LATAM and worldwide, servicing by two principal sea ...

based inOaks, PENNSYLVANIA (USA)
VascuTech Medical, LLC has evolved from a consulting firm serving major customers such as C.R. Bard, to a contract manufacturer of FDA and ISO 13485 registered medical device manufacturer serving OEMs and healthcare providers in interventional ...
Dialysis presents a unique set of vascular access challenges from clotting to infection. VascuTech Medical is a leading contract manufacturer of clinical solutions for acute, chronic and peritoneal dialysis ...

based inTraverse City, MICHIGAN (USA)
Thompson Surgical Instruments delivers Uncompromised Exposure to a variety of surgical specialties with the Thompson Retractor. In use for over fifty years, the Thompson Retractor is the only mechanical retractor to offer one frame for all exposure ...
The OneFrame Retractor Set by Thompson Surgical is a powerful tool designed for enhanced surgical access and retraction. It integrates seamlessly with the versatile OneFrame platform, delivering enhanced general and ...

based inDundee, UNITED KINGDOM
Vascular Flow Technologies (VFT) has evolved into an ISO 13485 Medical Device Development company with unique insight and capabilities in pre-clinical development and testing. VFT developed and patented its novel Spiral Laminar Flow™ (SLF) ...
The graft is an expanded polytetrafluoroethylene (ePTFE) graft with custom features injection moulded of Chronoflex polyurethane. The distal portion of the graft contains the patented Spiral Flow™ Inducer, the Inducer Indicator Ring and ...

based inEssen, GERMANY
Medical technology can be directed towards an indication or to everyone concerned. We orientate ourselves accordingly. Naturally, the main focus is on the patient, especially where port catheters are concerned. Port systems are intended for ...
TITAN-PORT APH (exctracorporeal apheresis) is a fully implantable port catheter system providing an access facility for performing extracorporeal apheresis. The set contains a titanium port chamber with a self-sealing silicone membrane and a ...

based inLatham, NEW YORK (USA)
AngioDynamics, with its disruptive and innovative medical devices, addresses unmet patient needs by elevating the standard of care for chronic and acute disease states in vascular, peripheral vascular, and oncology medicine. These devices provide ...
The “next generation” DuraFlow 2 chronic hemodialysis catheter from AngioDynamics continues to deliver the performance-tough features you demand and the care your patient deserves. Building on the standards for high performance, strength ...

based inDurham, NORTH CAROLINA (USA)
At 410 Medical our mission is to help save the lives of critically ill patients by developing innovative tools that assist healthcare providers in improving resuscitation. 410 Medical takes its name from the calculation of fluid delivery goals in ...
FAST Deliver 1 unit of whole blood in 2 minutes, PORTABLE Goes from field to hospital, EFFICIENT Easy one-handed operation, SIMPLE Handle and tubing packaged together for quick and easy ...

based inMidvale, UTAH (USA)
Utah Medical Products Inc, established in 1978, develops, manufactures and markets specialty medical devices recognized by healthcare professionals in Critical Care, Gynecology, Neonatology, Obstetrics, Surgery and Urology as the standard for ...
The Deltran Plus closed needleless arterial blood sampling system was designed to offer increased protection to both patients and healthcare workers. As a closed needleless system, Deltran® Plus gives the clinician the confidence of absolute ...

based inCoralville, IOWA (USA)
For more than 35 years, CIVCO Medical Solutions has been committed to making image-guided procedures safer through the design, manufacturing, and marketing of medical products. Our products are trusted by physicians and clinicians in radiology, ...

based inOakville, ONTARIO (CANADA)
CHS Ltd. (est. 1967) is a privately held medical device manufacturer and specialty distributor located in Oakville, Ontario. Customers served are in the acute hospital and non-acute healthcare space in Canada and Internationally. CHS ...

based inDeerfield, ILLINOIS (USA)
Baxter was founded in 1931, however our history doesn't have one starting point. Our commitment to addressing unmet needs was built on the shoulders of healthcare pioneers whose work resulted in first-of-its-kind innovations in IVs, renal care, ...
The Oxiris set is available in select countries around the world. Oxiris is the only blood purification filter set to effectively remove inflammatory mediators, endotoxin, fluid, and uremic toxins simultaneously. ...

based inWallingford, CONNECTICUT (USA)
QuikClot Hemostatic Products provide a bleeding control system that addresses bleeding scenarios from abrasions to traumatic injuries.25,26,27 Its proprietary hemostatic technology consists of a non-woven material impregnated with kaolin, an ...

based inMorrisville, NORTH CAROLINA (USA)
Teleflex is a global provider of medical technologies designed to improve the health and quality of people’s lives. We apply purpose driven innovation – a relentless pursuit of identifying unmet clinical needs – to benefit patients and healthcare ...

based inFaridabad, INDIA
Founded in 2007, Newtech Medical Devices is Delhi-NCR, India based one of the leading manufacturing Company with CE and ISO compliance. Newtech develops, manufactures, markets & supports cutting edge vascular access devices and accessories to meet ...

based inSalt Lake City, UTAH (USA)
At Health Line, our mission is to be one of the world’s leading manufacturers of vascular access, infusion therapy, and infection control products. We are committed to upholding strong quality management and regulatory systems to meet the demands of ...
The ORION™ II CT Central Venous Catheters are power injectable and intended for short-term infusion therapy. Available in a variety of sizes and lengths, the ORION™ II has an indwelling central line indicated for pressure injection of ...

based inLake VIlla, ILLINOIS (USA)
SAI was founded in 1995 with the goal of helping researchers develop and implement reliable infusion techniques. We want infusion and sampling to be low-stress for both the researcher and the subject. Throughout our 20+ years, we have become known ...
Our safe and secure rat Catheter Access Buttons™ feature CannuLock™ technology for the best catheter fit. The smooth barbed connector locks with a catheter to eliminate the risk of slipping or disconnecting. You may also like ...

based inDongguan, CHINA
Founded in 2012, BrosMed Medical is a leading medical device multinational company specializing in R&D, production and global sales of high-performance intravascular interventional medical devices and accessories. BrosMed has more than 40 product ...

based inBirmingham, ALASKA (USA)
Endomimetics has developed a BioNanomatrix that can be used to coat these devices and mimic human tissue which minimizes the need for additional surgeries, improves the longevity of the device, reduces complications associated with having foreign ...
In many medical settings, devices are commonly used to help doctors treat patients. For example, stents are often used to open blocked arteries, repair torn vessels, and even to keep nasal passages from swelling shut after surgery. Unfortunately ...