- Home
- Companies
- north america
- antigen testing
Show results for
Refine by
Antigen Testing Suppliers In North America
38 companies found

based inEast Brisbane, AUSTRALIA
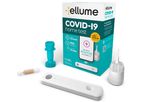
Ellume is a digital diagnostics company that develops, manufactures, and commercializes high-performance, connected products for healthcare professionals and consumers. It is at the forefront of accurate, rapid and accessible testing that is ...
The Ellume COVID-19 Home Test is an over-the-counter, rapid self-test which detects SARS-CoV-2 antigen. The test is suitable for ages 2 years and above for over-the-counter home use ...

based inShenzhen, CHINA
The company will continue to regard integrity as the most important cultural spirit; in the whole process of business decision-making and management, we abide by the principle of 'quality of products, quality service' and 'service for human health'; ...

based inIrvine, CALIFORNIA (USA)
Established in 2011, Pathnostics is driven to pioneer a better future for physicians, patients, and the healthcare system with solutions that transform diagnostic and therapeutic dilemmas. Core capabilities in infectious disease and cancer ...
The Pathnostics Respiratory Pathogens Test is a highly specific and sensitive multiplex PCR for the nucleic acid detection of SARS-Cov-2, Influenza A, Influenza B, or respiratory syncytial virus (RSV). Using one nasal swab, our Respiratory Pathogens ...

based inBethlehem, PENNSYLVANIA (USA)
OraSure Technologies empowers the global community to improve health and wellness by providing access to accurate, essential information. Together with its wholly-owned subsidiaries, DNA Genotek, Diversigen, and Novosanis, OraSure provides its ...
The OraQuick® Ebola Rapid Antigen Test has been granted De Novo 510(k) clearance by the U.S. Department of Health and Human Services for the following intended use: The OraQuick® Ebola Rapid ...

based inHayward, CALIFORNIA (USA)
Novodiax is dedicated to advancing tissue-based diagnostics and immunoassays through innovation. Our signature technology platform is a proprietary polymer-based detection system. Based on this technology we have developed a series of highly ...
Cluster of Differentiation 20 (CD20) is a polymerized horseradish peroxidase (pHRP)-labeled anti-CD20 (R272) rabbit monoclonal antibody intended for laboratory use using frozen or formalin-fixed-paraffin-embedded tissues. CD20 also known as ...

based inIndianapolis, INDIANA (USA)
MiraVista Veterinary Diagnostics is the veterinary division of MiraVista Diagnostics, a specialty laboratory in Indianapolis, Indiana devoted to detection and monitoring of systemic fungal infections. The company was founded in 2002 by the renowned ...
Clinical Significance: The Aspergillus Antigen EIA test aids in the diagnosis of systemic aspergillosis in animals. This test has low sensitivity in dogs with sino-nasal aspergillosis; therefore, ...

based inSarasota, FLORIDA (USA)
Lumos Diagnostics specializes in rapid, cost-effective and complete point-of-care (POC) diagnostic test solutions to help healthcare professionals more accurately diagnose and manage medical conditions. Lumos offers customized assay development and ...
CoviDx™ SARS-CoV-2 Rapid Antigen Test is a lateral flow assay intended for the qualitative detection of the nucleocapsid protein antigen from SARS-CoV-2 in nasal swabs and nasopharyngeal swabs ...

based inSomerset, NEW JERSEY (USA)
Even the most sophisticated, cutting-edge medical diagnostics aren’t of value unless we can get them to those who need them most. At Access Bio, we believe every life is precious, and every human on earth deserves the opportunity to live a healthy ...
The CareStart™ COVID-19 Antigen Test is a lateral flow immunochromatographic assay intended for the qualitative detection of the nucleocapsid protein antigen from SARS-CoV-2 in nasopharyngeal ...

based inGlen Allen, VIRGINIA (USA)
GENETWORx is dedicated to improving the lives of all people, whether it’s through COVID-19 diagnostic or antibody testing, or pharmacogenomics. GENETWORx Laboratory is a health management service company that specializes in bringing the benefits of ...
Antigen tests detect proteins specific to Coronavirus within 15 minutes. These tests are quite convenient, but not as universally accurate as the PCR tests. They are generally ...

based inRancho Dominguez, CALIFORNIA (USA)
DxTerity is a patient-centric genomics company passionate about putting patients with immune-mediated diseases at the center of everything that we do. We’re harnessing the power of real-world genomics with convenient from-home RNA monitoring to help ...
The DxTerity SARS-CoV-2 (COVID-19) RT-PCR CE saliva-based test provides the convenience of at-home sample collection with results available within 24-72 hours. Our highly-sensitive, gold-standard PCR test has been verified to work with all COVID-19 ...

based inResearch Triangle Park, NORTH CAROLINA (USA)
Galaxy Diagnostics was launched in 2009 to commercialize a more sensitive test method for Bartonella species infection, called Bartonella ePCR. The underlying technology, developed by Galaxy cofounders, Drs. Ed Breitschwerdt and Ricardo Maggi at NC ...
A revolutionary test method to confirm active Borrelia infection. This urine-based test provides more sensitive direct detection for Borrelia burgdorferi infection at all stages of Lyme disease than standard ...

based inSan Diego, CALIFORNIA (USA)
REAGEN LLC, focusing on ELISA Kits including clinical disease detecting kits, food & feed safety test kits and animal epidemic virus detecting kits in US for 11 years. REAGEN produces innovative diagnostic system for detections of estrogens, ...
Contact Angela: reagenllc@gmail.com Principle: Sandwich lateral flow immunochromatographic assay Intended use: Detection of canine distemper virus antigen Specimen: Ocular and nasal secretions, serum, plasma Shelf life: 24 months ...

based inVancouver, BRITISH COLUMBIA (CANADA)
Response Biomedical has a global distribution network and is focused on enabling our strong network of partners to be leaders in their markets by providing exceptional levels of service and support. Response Biomedical fosters a values-based ...
Our all-in-one RAMP® COVID-19 Antigen kit offers on the spot actionable results you can trust. Combined with our RAMP® 200 instrument, we offer rapid triage of nasal swab samples in 15 minutes. ...

based inRockville, MARYLAND (USA)
Founded in 2005, Maxim Biomedical (MaximBio) is a leading diagnostic healthcare company enhancing global health through the development and manufacture of trusted diagnostic solutions. Our mission is to create diagnostic products that are ...
The MaximBio ClearDetect™ COVID-19 Antigen Home Test is an antigen test that allows you to detect proteins from the virus that causes COVID-19. Made easy - An ...

based inSpringdale, ARKANSAS (USA)
NOWDiagnostics Inc., is a leader in innovative diagnostics testing. Dr. Qinwei Shi helped revolutionize lateral flow assay testing using very small sample sizes. Dr. Shi developed and perfected the platform which now is the foundation for all our ...
The ADEXUSDx HIV 1/2 Test is an immunoassay used for the qualitative detection of antibodies to human immunodeficiency virus in whole ...

based inVancouver, BRITISH COLUMBIA (CANADA)
We’re a leading bioscience company focused on accelerating new and innovative medical solutions for positive impact. XPhyto is striving to have a positive impact on global health. We are creating potentially life-changing solutions through research, ...
XPhyto Therapeutics is focused on helping economies restart and businesses get up and running to bring life back to normal with a rapid, accurate and cost effective COVID ...

based inSan Diego, CALIFORNIA (USA)
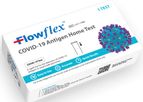
For over 23 years, ACON has led the way in making high quality diagnostic and medical devices more affordable to people all around the world. Headquartered in San Diego, California, the US office is the center of strategic management, ...
Critical Information When & Where You Need It; The Flowflex™ COVID-19 Antigen Home Test is all you need to determine your family’s Covid-19 status, whether symptoms are present or not. Can be used on ...

based in, MASSACHUSETTS (USA)
MtoZ Biolabs is an integrate contract research organization (CRO) providing advanced proteomics, metabolomics, bioinformatics, and biopharmaceutical analysis services to researchers in biochemistry, biotechnology, and biopharmaceutical fields. The ...

based inJamnagar, INDIA
Spark Mobile Diagnostics was conceived with a multi-year research inspired by the technology revolution bought by development of paper-based affordable diagnostics. We believe that mobile phone penetration across the globe provides a tremendous ...
CoV-Check DIRECT Covid-19 Antigen Test is a rapid immunochromatographic assay that utilizes specific monoclonal antibodies to detect Nucleocapsid protein of SARS-CoV-2 virus in nasopharyngeal swab specimens. ...

based inSanta Clara, CALIFORNIA (USA)
LumiQuick Diagnostics Inc., located in the heart of Silicon Valley, California, USA, develops, manufactures and markets high quality point of care tests and other immunoassay kits for the world-wide In vitro diagnostic market. Through years of ...
Malaria pf/pv Antigen Test Card is an in vitro qualitative immunochromatographic assay for the rapid detection and differentiation of Malaria P. falciparumspecific histidine rich protein-2 (Pf HRP-2) and Malaria P. ...