Show results for
Refine by
Locations
- USA
- Alabama
- Alaska
- Arizona
- Arkansas
- California
- Colorado
- Connecticut
- Delaware
- District Of Columbia
- Florida
- Georgia (USA)
- Hawaii
- Idaho
- Illinois
- Indiana
- Iowa
- Kansas
- Kentucky
- Louisiana
- Maine
- Maryland
- Massachusetts
- Michigan
- Minnesota
- Mississippi
- Missouri
- Montana
- Nebraska
- Nevada
- New Hampshire
- New Jersey
- New Mexico
- New York
- North Carolina
- North Dakota
- Ohio
- Oklahoma
- Oregon
- Pennsylvania
- Rhode Island
- South Carolina
- South Dakota
- Tennessee
- Texas
- Utah
- Vermont
- Virginia
- Washington
- West Virginia
- Wisconsin
- Wyoming
Antigen Testing Suppliers In USA
33 companies found

based in, MASSACHUSETTS (USA)
MtoZ Biolabs is an integrate contract research organization (CRO) providing advanced proteomics, metabolomics, bioinformatics, and biopharmaceutical analysis services to researchers in biochemistry, biotechnology, and biopharmaceutical fields. The ...

based inSpringdale, ARKANSAS (USA)
NOWDiagnostics Inc., is a leader in innovative diagnostics testing. Dr. Qinwei Shi helped revolutionize lateral flow assay testing using very small sample sizes. Dr. Shi developed and perfected the platform which now is the foundation for all our ...
The ADEXUSDx HIV 1/2 Test is an immunoassay used for the qualitative detection of antibodies to human immunodeficiency virus in whole ...

based inSan Diego, CALIFORNIA (USA)
REAGEN LLC, focusing on ELISA Kits including clinical disease detecting kits, food & feed safety test kits and animal epidemic virus detecting kits in US for 11 years. REAGEN produces innovative diagnostic system for detections of estrogens, ...
Contact Angela: reagenllc@gmail.com Principle: Sandwich lateral flow immunochromatographic assay Intended use: Detection of canine distemper virus antigen Specimen: Ocular and nasal secretions, serum, plasma Shelf life: 24 months ...

based inNanjing, CHINA
Founded in 2002, Jiji Biotech Co., Ltd.is a medical and health industry chain that integrates independent research and development, large-scale production and professional marketing. It has many subsidiaries and offices at home and abroad. the ...
Test Item: SARS-CoV-2 Antigen. Sample Type: Human nasal swab. Test Time: 15 min. Methodology: Immunofluorescence ...

based inResearch Triangle Park, NORTH CAROLINA (USA)
Galaxy Diagnostics was launched in 2009 to commercialize a more sensitive test method for Bartonella species infection, called Bartonella ePCR. The underlying technology, developed by Galaxy cofounders, Drs. Ed Breitschwerdt and Ricardo Maggi at NC ...
A revolutionary test method to confirm active Borrelia infection. This urine-based test provides more sensitive direct detection for Borrelia burgdorferi infection at all stages of Lyme disease than standard ...

based inBethlehem, PENNSYLVANIA (USA)
OraSure Technologies empowers the global community to improve health and wellness by providing access to accurate, essential information. Together with its wholly-owned subsidiaries, DNA Genotek, Diversigen, and Novosanis, OraSure provides its ...
The OraQuick® Ebola Rapid Antigen Test has been granted De Novo 510(k) clearance by the U.S. Department of Health and Human Services for the following intended use: The OraQuick® Ebola Rapid ...

based inSan Diego, CALIFORNIA (USA)
We apply cutting-edge science to create innovative therapies that will improve the lives of those who suffer from cancer, intractable pain and COVID-19. Cancer is genetically diverse, highly adaptive, constantly mutating and virtually invisible to ...
SIMPLE: 3-step test procedure. RAPID: Produces Results in 15 minutes or less. CONVENIENT: Nasal swab with simple 3 step instructions and visible read. ACCURATE: Highly sensitive platinum colloid-based lateral flow ...

based inSanta Maria, CALIFORNIA (USA)
Hardy Diagnostics is a 100% employee-owned company that is licensed by the FDA as a medical device manufacturer, and its quality management system is ISO 13485 certified. Hardy Diagnostics has been helping people live healthier lives since 1980. Our ...
An immunochromatographic rapid diagnostic test (RDT) intended for the qualitative detection of nucleocapsid protein antigen from SARS-CoV-2 in direct nasopharyngeal (NP) or anteriornasal (AN) swab specimens. ...

based inNew Orleans, LOUISIANA (USA)
Our mission is to advance infectious disease diagnosis and improve patient care using novel diagnostic platforms that produces rapid and accurate results from patient samples. Our versatile platforms address critical needs for current infectious ...
To address these issues, NanoPin uses an antibody enrichment approach to capture species-specific biomarker peptides released upon trypsin digestion of the serum of infected individuals and employs mass spectrometry to identify these peptides with ...

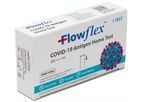
based inUnion City, TENNESSEE (USA)
Mountain Horse Solutions is a leading provider of innovative and high-quality operational equipment, supplies, technology, and training necessary to support the performance of mission-critical teams around the globe. We provide superior customized ...
The Flowflex COVID-19 Antigen Home Test is a rapid test for the detection of SARS-CoV-2. The lateral flow chromatographic immunoassay is intended for the qualitative detection of the nucleocapsid ...

based inSarasota, FLORIDA (USA)
Lumos Diagnostics specializes in rapid, cost-effective and complete point-of-care (POC) diagnostic test solutions to help healthcare professionals more accurately diagnose and manage medical conditions. Lumos offers customized assay development and ...
CoviDx™ SARS-CoV-2 Rapid Antigen Test is a lateral flow assay intended for the qualitative detection of the nucleocapsid protein antigen from SARS-CoV-2 in nasal swabs and nasopharyngeal swabs ...

based inSomerset, NEW JERSEY (USA)
Even the most sophisticated, cutting-edge medical diagnostics aren’t of value unless we can get them to those who need them most. At Access Bio, we believe every life is precious, and every human on earth deserves the opportunity to live a healthy ...
The CareStart™ COVID-19 Antigen Test is a lateral flow immunochromatographic assay intended for the qualitative detection of the nucleocapsid protein antigen from SARS-CoV-2 in nasopharyngeal ...

based inDavis, CALIFORNIA (USA)
Cell Technology was founded in 1998 to provide research solutions for the academic, biotechnology and pharmaceutical industries. Our focus is to develop unique assays that allow researchers to study cellular functions using cell-permeable reagents. ...

based inPoway, CALIFORNIA (USA)
Alfa Scientific Designs, Inc. is a California-licensed, cGMP compliant, and ISO 13485 registered manufacturer of in-vitro diagnostic devices. Founded in 1996, ALFA is a leader in the industry in developing and manufacturing high quality rapid ...

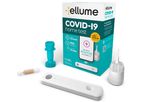
based inEast Brisbane, AUSTRALIA
Ellume is a digital diagnostics company that develops, manufactures, and commercializes high-performance, connected products for healthcare professionals and consumers. It is at the forefront of accurate, rapid and accessible testing that is ...
The Ellume COVID-19 Home Test is an over-the-counter, rapid self-test which detects SARS-CoV-2 antigen. The test is suitable for ages 2 years and above for over-the-counter home use ...

based inRancho Dominguez, CALIFORNIA (USA)
DxTerity is a patient-centric genomics company passionate about putting patients with immune-mediated diseases at the center of everything that we do. We’re harnessing the power of real-world genomics with convenient from-home RNA monitoring to help ...
The DxTerity SARS-CoV-2 (COVID-19) RT-PCR CE saliva-based test provides the convenience of at-home sample collection with results available within 24-72 hours. Our highly-sensitive, gold-standard PCR test has been verified to work with all COVID-19 ...

based inMissoula, MONTANA (USA)
FYR Diagnostics is developing a powerful portfolio of biomarker detection technologies with a focus on nucleic acid amplification. Our mission is to commercialize and implement these novel technologies to enable the accelerated prediction, ...

based inLimerick, PENNSYLVANIA (USA)
Rockland Immunochemicals, Inc. offers a robust catalog of antibodies to suit any project. No matter the context—basic research to disease therapy, phosphorylated to methylated, and beyond—Rockland embraces the challenge to design, produce, validate, ...
CD45 is encoded by the PTPRC (Protein tyrosine phosphatase receptor type C) gene. CD45, also known as the leukocyte common antigen (LCA), is a member of the protein tyrosine phosphatase (PTP) family. It is present on all human leukocytes including ...

based inHouston, TEXAS (USA)
We are a CLIA high complexity laboratory and a developer and manufacturer of blood diagnostic tests. We specialize in molecular testing and Point of Care immunoassay. At Accel Diagnostics our mission is to transform the way a wide variety of medical ...
Our quantitative SARS-CoV-2 (COVID-19) IgG antibody test is designed to measure the level of the IgG antibodies against the Spike protein of SARS-COV-2 virus - the virus that causes COVID-19. Unlike most antibody tests, which provide a ...

based inIndianapolis, INDIANA (USA)
MiraVista Veterinary Diagnostics is the veterinary division of MiraVista Diagnostics, a specialty laboratory in Indianapolis, Indiana devoted to detection and monitoring of systemic fungal infections. The company was founded in 2002 by the renowned ...
Clinical Significance: The Aspergillus Antigen EIA test aids in the diagnosis of systemic aspergillosis in animals. This test has low sensitivity in dogs with sino-nasal aspergillosis; therefore, ...