Refine by
Artery Disease Suppliers Serving Congo
58 companies found

based inCarlsbad, CALIFORNIA (USA)
CeloNova BioSciences, Inc. is developer and manufacturer of a portfolio of novel surfaced coated technologies, including the proprietary Polyzene-F nanocoating. This next-generation nanocoating is the result of years of rigorous scientific research ...
Our proprietary COBRA SG drug-coated PTCA balloon includes a bio-active medical coating designed to improve adhesion of organic and inorganic substrates and optimize drug delivery. Developed at a leading, international medical university, the novel ...

based inGeleen, NETHERLANDS
Promicol was established in 2000, founded on many years of experience in all aspects of ATP technology. We are based in The Netherlands, centrally located in Europe, with excellent transport facilities and logistic services. We provide systems that ...

based inWhite Bear Lake, MINNESOTA (USA)
Nextern is a collaborative medical device company. We bring an unmatched core competence in medical device product design and development through manufacturing as a leverageable asset to collaborators like you, who are focused on accelerating the ...
We have expertise in producing microcatheters used in neurovascular, cardiovascular, and peripheral vascular applications. Our goal is to design and manufacture innovative products that provide better patient outcomes and advance the field of ...

based inRedondo Beach, CALIFORNIA (USA)
Quantimetrix designs, develops, and manufactures laboratory quality products at our headquarters in Redondo Beach, California. We are leaders in the field of liquid-stable quality control products, having pioneered these innovations more than 40 ...

based inIrvine, CALIFORNIA (USA)
Walk Vascular was founded in 2010, with a mission to treat and transform the lives of patients suffering from peripheral arterial and venous disease. Its JETi Thrombectomy System (JETi) is manufactured within The Company's 19,000 sq. ft., Irvine, ...

based inMonmouth Junction, NEW JERSEY (USA)
MedChemExpress (MCE) offers a wide range of high-quality research chemicals and biochemicals (novel life-science reagents, reference compounds and natural compounds) for scientific use. We have professionally experienced and friendly staff to meet ...
Trimetazidine-d8 (dihydrochloride) is the deuterium labeled Trimetazidine dihydrochloride. Trimetazidine dihydrochloride is a selective long chain 3-ketoyl coenzyme A thiolase inhibitor with an IC50 of 75 nM, which can inhibit β-oxidation of free ...

based inSão Paulo, BRAZIL
We were born on July, 15, 1994 in Amparo/SP, and the essence of our company is to use state-of -the-art technology to develop equipment for human wellness, in physical rehabilitation, esthetics or esthetic medicine. Today, we are located in a built ...

based inSoucieu en Jarrest, FRANCE
We are committed to combine sustainable growth with innovation and creativity to provide high-quality solutions, expertly made in France, and used all over the world. We have been specialized in the design, manufacture and sales of transcranial ...
Atys has been manufacturing devices for the diagnosis of peripheral vascular disease (peripheral arterial disease (PAD) and venous insufficiency) for 30 ...

based inBrea, CALIFORNIA (USA)
Whatever comes next, we make sure life comes first. For health care professionals working with critically ill patients, life isn’t a given—it’s the ultimate goal. They face demanding, heartbreaking and rewarding situations every day. Their goal is ...
Transcutaneous monitoring tailored to your needs. Reveal wound healing capacity with the PeriFlux 6000 tcpO2 stand-alone transcutaneous monitor. PeriFlux 6000 tcpO2 stand-alone transcutaneous monitor can support you in assessing patients with lower ...

based inAndover, MASSACHUSETTS (USA)
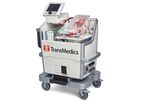
TransMedics was founded to address the growing need for healthier organs for transplantation. We are focused on transforming the standard of care - increasing organ utilization, improving patient outcomes, and reducing transplant costs. The Organ ...
The Only Heart Device Currently Under FDA Review for Utilized and Unutilized Donor Organs. The OCS Heart is a portable, warm perfusion, and monitoring system designed to keep a donor heart at a human-like, metabolically active state. By monitoring ...

based inLeeds, UNITED KINGDOM
Founded in 2009, Leeds-based Arterius is developing a next-generation bioresorbable cardiovascular scaffold (stent) with an emerging market-leading clinical profile. The founding directors have significant experience in the development of medical ...

based inJarfälla, SWEDEN
Perimed AB is a Swedish company, founded in 1981 and headquartered just outside Stockholm, that develops, manufactures and markets state-of-the-art diagnostic equipment to assess microcirculation and peripheral perfusion as well as peripheral ...
Our sensitive laser Doppler technique provides accurate toe pressure measurements. By adding the SPP technique, a more comprehensive wound healing assessment can be obtained. All pressure tests are easy and fast to perform thanks to our ...

based inJeddah, SAUDI ARABIA
Gulf Medical Company was founded in 1983 as a medical supply company specialized in the sales and marketing of medical devices and equipment, with the sole proprietorship of the vision “To bring the most innovative state of the art medical ...

based inSanta Clara, CALIFORNIA (USA)
Shockwave is focused on developing and commercializing products intended to transform the way calcified cardiovascular disease is treated. Shockwave aims to establish a new standard of care for the interventional treatment of atherosclerotic ...
Neovasc is focused on creating better outcomes for difficult-to-treat cardiology patients through the development of two novel products: Reducer™* for treatment of refractory angina and the Tiara™ System* for treatment of mitral valve ...

based inBonn, GERMANY
CARDIONOVUM, a medical technology company based in Bonn, Germany, develops and commercializes innovative medical devices such as the new generation of DCB and drug-eluting stents for the treatment of coronary and peripheral artery disease. The ...
The LEGFLOW balloon drug coating quality is a major step beyond contemporary first and second generation DCB ...

based inBurlington, MASSACHUSETTS (USA)
LeMaitre is a leading global innovator, manufacturer and marketer of devices for the treatment of peripheral vascular disease. We’re proud to provide vascular surgeons with the solutions they need to diagnose and heal their patients. While LeMaitre ...

based inChampionsGate, FLORIDA (USA)
Our purpose is to improve health of the patients all around the world with our strong, hardworking, dedicated and clinically driven biomedical solutions team. Our products and technologies are mostly focused on endovascular treatment solutions with ...
is a coronary balloon-expandable Stainless Steel stent system consisting of everolimus particles. It is indicated for de novo and in-stent restenotic lesions in patients with coronary artery disease – ...

based inVochov, CZECH REPUBLIC
EMBITRON Ltd. is a recognized producer and supplier of medical devices for physiotherapy, medical rehabilitation, and other medical fields. During more than twenty years of its track record, EMBITRON Ltd. has become one of the major suppliers of ...
Blood circulation in limbs. Unique instrument for vacuum compression therapy. Effective treatment of disorders of blood flow to the extremities, stagnant metabolism, and impaired trophic ...

based inSan Jose, CALIFORNIA (USA)
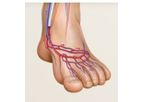
LimFlow was founded with the deep belief that more could be done for the millions of patients worldwide suffering from the most severe form of peripheral artery disease — Chronic Limb-Threatening Ischemia (CLTI) — and who are facing the prospect of ...
Living with Peripheral Artery Disease (PAD) that progresses to CLTI is challenging. Life revolves around chronic leg pain that can become unbearable over time. The end stage of the disease is ...

based inFederal Way, WASHINGTON (USA)
Established in 1987, DiagnosTechs was the first laboratory to introduce saliva hormone testing into routine clinical practice. In 1995, DiagnosTechs added saliva and stool-based gastrointestinal and food sensitivity testing, which expanded the level ...