Refine by
Specimen Collection Suppliers Serving Congo
58 companies found

based inGuangzhou, CHINA
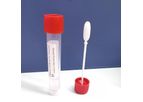
Guangzhou Improve Medical Instruments Co., Ltd (hereinafter referred to as “Improve Medical”), established in 1996, is a high-tech enterprise which provides relevant technologies, products and services for Clinical Laboratory and Clinical Nursing. ...
IMPROVACUTER® ESR Tubes are used for Westergren sedimentation rate determination. The tubes contain buffered sodium citrate solution as an anticoagulant. Citrate concentration of either 0.109M(3.2%) or 0.129M (3.8%) is available. The mixing ...

based inCarlsbad, CALIFORNIA (USA)
Coretests products has been certified by ISO 9001, ISO 13485, CE, and FDA. Our team has develop numerous products, ranging from proteins, rapid tests, to PCR and POCT products. The hundreds of products fall under five categories, Fertility, Drug of ...
Product Name : One Step Fecal Occult Blood Test Device. Accessories : Instruction for use, feces collection tube. Principle of Test : Immunochromatographic assay. Product Performance:Sensitivity : 50 ...

based inFrederick, MARYLAND (USA)
In 2019, ProMedDx and GLAS Bio were acquired by Precision for Medicine, part of Precision Medicine Group. This new combination significantly bolstered the selection, scientific prowess and solutions available to customers from a single partner. With ...
Extensive inventory of pathologist-reviewed formalin-fixed paraffin-embedded tissue blocks & slides from normal and diseased subjects. ...

based inCommack, NEW YORK (USA)
Crystalgen was founded from the acquisition of a small startup company in 1994, currently we are a global manufacturer and supplier of high quality laboratory essentials, biochemicals, and reagents. At Crystalgen we focus on innovation and ...
Total Length 155mm. Tip Width 5mm. Tip Thickness 5mm. Tip Length 16mm. Handle Diameter 2.5mm. Breakpoint 81mm. ABS handle, Nylon Fiber tip. Sterile, Individually Wrapped. ...

based inBeaverton, OREGON (USA)
Sedia Biosciences is a privately held Delaware-registered medical device and diagnostics company founded in 2009 and based in Beaverton, Oregon, USA. Our commitment is to become a leader in three areas intended to advance global access to care: ...
Designed to conveniently collect, preserve, and transport oral specimens containing DNA. ...

based inRHO - (MILAN), ITALY
STERYLAB has devoted its efforts to the production of medical needles and devices for over 45 years, keeping its development in step with the constant progress in medicine and the latest advances in technology. Due to the precision workmanship of ...
The design of BEST-LISAS® is the most advanced to date and guarantees to the physician the following ...

based inDavis, CALIFORNIA (USA)
Gold Standard Diagnostics provides comprehensive diagnostic solutions that improve lab efficiency and minimize overall costs. Through an extensive product menu, automated instrument platforms, and outstanding customer service, we solve real ...
The Antigen Rapid Test assay from the Gold Standard Diagnostics Group is a rapid chromatographic immunoassay for the qualitative detection of SARS-CoV-2 Nucleocapsid (N) Protein antigens present in respiratory ...

based inPalo Alto, CALIFORNIA (USA)
Nirmidas Biotech was founded in September of 2013. We are a member of the Janssen Labs (JLABS) and QB3 programs, the Stanford StartX Med Program, and the MIT Industrial Liaison Program (MIT ILP). We are also a StartX Med COVID-19 Task Force Company ...
Nirmidas Biotech Inc. has developed an antigen test aiming for FDA EUA, MidaSwift™ SARS-CoV-2 Antigen Detection Kit, for the qualitative detection of the Nucleocapsid antigen from SARS-CoV-2. The test is curently for Research Use Only (RUO), ...

based inWuxi, CHINA
Bioteke Corporation, which focuses on molecular diagnostics and rapid detection, particularly, on nucleic acid extraction, is a professional high-tech manufacturer. It is set in Wuxi City, Jiangsu province, China. By 2022, Bioteke has dozens of ...
Bioteke disposable specimen collection kit ST6001 is intended for collection , transportation and storage of specimen, ensure the stability of nucleic acid (DNA/RNA) in samples. ...

based inVancouver, WASHINGTON (USA)
Oasis Diagnostics® Corporation was founded to conceive, develop, and commercialize high-quality tools for the non-invasive diagnosis and detection of diseases based on non-invasive specimens, particularly saliva or oral fluid. The Company’s ...
SimplOFy™ is a new kit for the standardized collection of 2.0 mL of whole saliva in the simplest way possible. The sample is immediately preserved by a dried stabilizing solution coated to the inside of the tube cap, which ...

based inOsaka, JAPAN
Alfresa Pharma develops, manufactures, and sells diagnostic reagents and equipments used in hospital laboratories and group medical examinations, with the mission of “delivering unprecedented new in vitro diagnostics.” We have applied our original ...
The conventional fecal calprotectin test often uses the ELISA method. However, our NESCAUTO® Cp Auto products use a colloidal gold agglutination method, which is an immunoassay. The gold colloid agglutination method optically measures the color ...

based inBrescia, ITALY
Since 1979 we explore all the fields of Microbiology, leveraging our Innovation by Togetherness approach to deliver top-notch solutions. Whether you’re elevating your Clinical Lab to the next level, searching for a custom collection and transport ...
Copan Universal Transport Medium (UTM®) system is intended for the collection, transport, and preservation of clinical specimens containing Viruses, Chlamydia, Mycoplasma, and ...

based inRoseto degli Abruzzi (TE), ITALY
Since 1983 Liofilchem has committed its Research and Development department to the production of reliable and high quality bacteriology products. The dedication to Customer satisfaction and exceptional manufacturing versatility have granted ...

based inChengdu, CHINA
Established in 1998, Chengdu Rich Science Industry Co., Ltd is a manufacturer specialized in research and development, production, sales and service of Medical Equipment, Medical Polymer Materials, Biotechnology, and Information Technology ...

based inMarlborough, MASSACHUSETTS (USA)
Hologic, Inc. is a medical technology company primarily focused on women’s health; it sells medical devices for diagnostics, surgery, and medical imaging. We’re an innovative medical technology company whose purpose is to enable healthier lives ...
The emergence of SARS-CoV-2 was unforeseen, and the worldwide outbreak of COVID-19 has already impacted millions. In response to the desperate public need for rapid high-volume testing we developed SARS-CoV-2 assays on both our Panther® and ...

based inZürich, SWITZERLAND
BC Platforms is a global leader in providing a powerful data and technology platform for personalized medicine and drug development, accelerating the translation of insights into clinical practice. Our technology drives the infinite loop between ...
Single solution for managing an end-to-end biobank process from participant consent to data and specimen ...

based inTikva, ISRAEL
LabOS is a sophisticated, AI-driven Laboratory Information System (LIS) delivered through the SaaS (Software-as-a-Service) model. It’s designed to serve a wide range of lab environments—clinical pathology, microbiology, anatomic pathology, and ...

based inMonmouth Junction, NEW JERSEY (USA)
MedChemExpress (MCE) offers a wide range of high-quality research chemicals and biochemicals (novel life-science reagents, reference compounds and natural compounds) for scientific use. We have professionally experienced and friendly staff to meet ...
Carprofen is a nonsteroid anti-inflammatory agent, acts as a multi-target FAAH/COX inhibitor, with IC50s of 3.9 μM, 22.3 μM and 78.6 μM for COX-2, COX-1 and FAAH, ...

based inSan Antonio, TEXAS (USA)
At Germaine Laboratories, we provide high-quality, reliable diagnostic testing supplies and services. We partner with our network of distributors to make sure we get the right product into the right hands at the right time. Browse our product ...
Our Drugs of Abuse Testing division has many years’ experience assisting major employers, School Districts, Criminal Justice and Drug Court entities throughout the United States. Our laboratory staff is dedicated to providing you with reliable ...

based inAmsterdam, NETHERLANDS
Self-screen has a long history and track record in translational HPV related science. The company was founded in 2008 to advance the implementation of scientific discoveries in the field of HPV related cancer screening into routine practice. Our ...
The QIAsure Methylation Test is intended to be used as a triage test for women with a positive HPV DNA test or for women with ASC-US cytology to identify those women who need to be referred for colposcopy. The QIAsure Methylation Test is a multiplex ...