- Home
- Companies
- equatorial guinea
- stent system
Refine by
Stent System Suppliers Serving Equatorial Guinea
47 companies found

based inHerzlia, ISRAEL
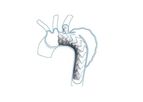
Privately held Endospan, headquartered in Herzlia (Tel Aviv), Israel, is a pioneer in the endovascular repair of aortic arch disease including aneurysms and dissections. Endospan’s NEXUSTM Aortic Arch Stent Graft System is the first endovascular ...
The NEXUS Aortic Arch Stent Graft System was specifically engineered for minimally-invasive total arch repair to overcome the challenges of the aortic arch anatomy. NEXUS is delivered by traveling through your ...

based inGyeonggi-do, SOUTH KOREA
Taewoong Medical Co., Ltd, was founded in 1991 and obtained CE approval for Niti-S™ brand products with excellence through product quality and technical expertise. We started business abroad with continuous growth and managed to obtain product ...
Lumen-apposing Metal Stent for the drainage of pancreatic pseudocyst or a gallbladder through a transgastric or transduodenal approach. Pre-loaded on the Electrocautery Delivery System. ...

based inBonn, GERMANY
CARDIONOVUM, a medical technology company based in Bonn, Germany, develops and commercializes innovative medical devices such as the new generation of DCB and drug-eluting stents for the treatment of coronary and peripheral artery disease. The ...
Unmatched XLIMUS clinical performance. The ultimate DES for complex artery lesions. XLIMUS is considered as the ultimate coronary DES stent system to treat complex coronary artery disease by reaching and crossing ...

based inShanghai, CHINA
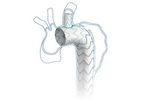
MicroPort is a global medical devices company, founded on the belief that every person has the right to high quality medical treatments and to live longer and healthier lives. All our solutions are designed to deliver on that belief. Our close ...
Serve the Arch Reserve the Branch: 0% mortality related dissection at 12 months. World's first branched aortic stent for clinical application. Saferand convenient method to reconstruct the left subclavian ...

based inReutlingen, GERMANY
It is probably a combination of many factors that has made us successful since 2004: the good collaboration of a medium-sized family company; the site in the heart of Germany’s medical technology region around the University of Tübingen; our ...
The new AndraBalloon represents the new generation of non-compliant PTA-Catheters optimized for dilation of large volume vessel and especially usefull as a delivery system for intervention with AndraStent. ...

based inSTAFFORD SPRINGS, CONNECTICUT (USA)
Hobbs Medical, is a privately held company founded in 1984, was among the first to specialize in the design and manufacture of custom diagnostic and therapeutic accessories for the Gastrointestinal and Pulmonary Endoscopy markets. Throughout the ...

based inEl Paso, TEXAS (USA)
Seisa Medical is a global full-service contract manufacturer of Class II and Class III medical devices and specialty components, ranging from implantable stents and tubing sets to pediatric and orthopedic-care products. We serve customers through ...

based inPembroke, MASSACHUSETTS (USA)
Founded in 1984, FCI stands as a global leader in the innovation and distribution of ophthalmic surgical devices. Originating in France, the company expanded its presence internationally by partnering with top surgeons across various markets. A ...

based inOldsmar, FLORIDA (USA)
MicroLumen has been a leading manufacturer of high performance medical products since 1987. Our high performance medical tubing is used in a wide range of minimally invasive, critical OEM applications such as cardiovascular catheters, stent delivery ...
Polyimide is a polymer thermoset plastic that has exceptional thermal stability, chemical resistance, and tensile strength. These characteristics make polyimide an ideal material for high-performance medical applications. The tubing is lightweight, ...

based inCarlsbad, CALIFORNIA (USA)
CeloNova BioSciences, Inc. is developer and manufacturer of a portfolio of novel surfaced coated technologies, including the proprietary Polyzene-F nanocoating. This next-generation nanocoating is the result of years of rigorous scientific research ...

based inNewport Beach, CALIFORNIA (USA)
Renata Medical is a company dedicated to innovating, developing, and marketing solutions for unmet pediatric medical needs. They have created a first-of-its-kind stent system, the Renata Minima Stent, which is FDA approved for treating vascular ...
Minima Infant Balloon-Expandable Stent System. 63 cm catheter working length, .014”/.018” guidewire ...

based inTel Aviv, ISRAEL
Medinol is dedicated to the science of cardiovascular intervention. For over 20 years, we have delivered over two million coronary stents to patients worldwide. Medinol is dedicated to the science of cardiovascular intervention. For over 20 years, ...
Flexx² is based on a tapered spring tip designed to eliminate tip flare out and buckling while enabling simultaneous flexibility and pushability. The entire coronary product line of Medinol builds on Flexx² catheters, including the EluNIR ...

based inDidcot, UNITED KINGDOM
Lombard Medical Limited is a UK-based medical device company focused solely on the minimally invasive treatment of aortic disease. In partnership with MicroPort Endovastec, we now offer the low-profile MinosTM AAA stent graft, the HerculesTM TAA ...
Furthering Treatment for Advance Pathologies. ...

based inTampa, FLORIDA (USA)
Concept Medical is driving innovation in medical devices, with a focus on developing technologies for drug delivery with a strong emphasis on intellectual property. Based in Tampa, Florida; Concept Medical has always striven to find solutions to ...
Drug is coated on the abluminal side only. Leading to mono directional drug release & less systemic exposure of drug which leads to faster ...

based inAhmedabad, INDIA
Advin Urology is a division of Advin Health Care, headquartered in Ahmedabad, India. Advin Urology division provides a complete range of Urology Surgery Products, Nephrology Products, Laparoscopy Products and Gynecology Products. Advin Urology ...
Double J stent with Pusher, suture and Guide Wire is available in one Tyvek Pouch as per EN-868-5 series standard. Double J Stent SET help to ...

based inChampionsGate, FLORIDA (USA)
Our purpose is to improve health of the patients all around the world with our strong, hardworking, dedicated and clinically driven biomedical solutions team. Our products and technologies are mostly focused on endovascular treatment solutions with ...
Atlas? is a coronary balloon-expandable Stainless Steel stent system consisting of everolimus particles. It is indicated for de novo and in-stent restenotic lesions in patients with coronary artery ...

based inJiangsu, CHINA
Kingshore New Resources ElectricGroup is a high-tech enterprise located in Economic Development Zone,Yangzhong, Jiangsu Province, which is consist of Kingshore New ResourcesCo., Ltd., Jiangsu Jin Di Electric Technology Co., Ltd.,Jiangsu Jinyuan ...

based inGalway, IRELAND
Advant Medical is a longstanding full stop solutions provider to the global medical device industry. We deliver an array of custom services from new product development to commercialisation for companies of all sizes. We are experts in Additive ...

based inHechingen, GERMANY
Bentley is an international medical technology company with headquarters in the South of Germany. At Bentley we are passionate about the development, manufacture and marketing of innovative products for the minimally invasive treatment of vascular ...
This peripheral stent graft system excels with a high radial force whilst maintaining extraordinary flexibility and a low profile. The cobalt-chrome stent platform is covered with a micro-porous ...

based inFaridabad, INDIA
Founded in 2007, Newtech Medical Devices is Delhi-NCR, India based one of the leading manufacturing Company with CE and ISO compliance. Newtech develops, manufactures, markets & supports cutting edge vascular access devices and accessories to meet ...
The Coherent Sirolimus eluting stent system with biodegradable polymer is a combination product comprised of two regulated components: a device (a coronary stent system) and a drug ...