Show results for
Refine by
Catheter Delivery Suppliers Serving Jordan
38 companies found

based inAdelaide, AUSTRALIA
Established in 2016 out of a desire to pursue optimal medtech solutions and to challenge the status quo, Numedico is a globally focused safety medical devices manufacturer and distributor based in Australia. We provide high quality medical supplies ...
This high quality retractable syringe is like no other on the market, and is available at a fraction of the price. ClickZip technology prevents reuse and needlestick injury due to our innovative titled needle technology that encapsulates the needle ...

based inReutlingen, GERMANY
It is probably a combination of many factors that has made us successful since 2004: the good collaboration of a medium-sized family company; the site in the heart of Germany’s medical technology region around the University of Tübingen; our ...
The semi-flexible hybrid cell Cobalt Chromium bare stent has been specifically designed for the effective treatment of Coarctation. Available in XL and ...

based inCarpinteria, CALIFORNIA (USA)
Our comprehensive technical capabilities range from the design and manufacture of minimally invasive, catheter, and handheld technology to the development and production of medical components utilizing advanced materials and processes. We lead the ...
Freudenberg Medical offers unsurpassable expertise in the design, development, and manufacture of minimally invasive surgical and catheter-based devices. Our product development and manufacturing teams collaborate to ensure design ...

based inCarlsbad, CALIFORNIA (USA)
CeloNova BioSciences, Inc. is developer and manufacturer of a portfolio of novel surfaced coated technologies, including the proprietary Polyzene-F nanocoating. This next-generation nanocoating is the result of years of rigorous scientific research ...

based inAlameda, CALIFORNIA (USA)
Penumbra, Inc., headquartered in Alameda, California, is a global healthcare company focused on innovative therapies. Penumbra designs, develops, manufactures and markets novel products and has a broad portfolio that addresses challenging medical ...
The Penumbra System MAX Reperfusion Catheters are intended for use in the revascularization of patients with acute ischemic stroke secondary to large vessel occlusion disease. The 3MAX and 4MAX Reperfusion Catheters feature advance tracking ...

based inWaltham, MASSACHUSETTS (USA)
Thermedical is developing a proprietary thermal ablation therapy that can treat significantly larger tissue volumes using a single device than conventional technology. Thermedical is a privately held medical device company based in Waltham, ...

based inWinsen, GERMANY
Stron Medical was launched in 2010 as a product line of QualiMed. With an impressive range of innovative products and a committed team of sales professionals, Stron Medical provides end-to-end solutions within Germany. QualiMed was found in 1997 as ...
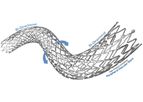
The POLARIS-ppADVANCE Peripheral Vascular Self-Expanding Stent System combines highly durable nitinol stent with a flexible delivery system to provide proven clinical ...

based inTampa, FLORIDA (USA)
Concept Medical is driving innovation in medical devices, with a focus on developing technologies for drug delivery with a strong emphasis on intellectual property. Based in Tampa, Florida; Concept Medical has always striven to find solutions to ...

based inHorsham, UNITED KINGDOM
Veryan has developed innovative technology to improve the performance of vascular stents by adopting the principle of biomimicry; developing structures that imitate those occurring naturally. Veryan’s vascular biomimetic stent technology involves ...
The Challenge: Peripheral Arterial Disease. People are living longer and with an increase in the incidence of obesity, diabetes and other contributing factors, claudication may cause substantial tissue changes and, in some scenarios, can lead to ...

based inAlpharetta, GEORGIA (US) (USA)
Spectrum is your first-call solutions provider for development through scaled manufacturing of critical polymer-based components and devices for medical and other demanding markets. We design solutions that support life-saving procedures and satisfy ...
With hundreds of materials and tools in stock and countless configurations readily available, we can rapidly produce prototypes to accelerate your extrusion, catheters, additive manufacturing, and 3D medical printing development ...

based inAliso Viejo, CALIFORNIA (USA)
In 1997 MicroVention was founded and incorporated in a small facility in Southern California. We are a worldwide community of over 2000 engineers, chemists, marketers, manufacturers, researchers, and innovators who share the same ethos - creating ...
Carotid Artery Stent Designed for Sustained Embolic ...

based inWinsen, GERMANY
QualiMed was found in 1997 as an OEM manufacturer for implantable medical devices with a focus on the development and regulatory approval of coronary stents and their respective delivery devices. Later the business was expanded to peripheral ...
The NAVALISADVANCE Peripheral Vascular Self-Expanding Stent System combines highly durable nitinol stent with a flexible delivery system to provide proven clinical ...

based inNashua, NEW HAMPSHIRE (USA)
We are the leader in micro manufacturing for the life sciences industry. We are a specialist in micro manufacturing, and we focus our technology on components and assemblies for medical device and diagnostics manufacturers. We’re built to solve ...
Equipped with the latest in technology advancements, our machines are capable of the most precise and fastest cutting speeds in the medtech industry. Our streamlined manufacturing facility is built to expedite the laser cutting process. All ...

based inIrvine, CALIFORNIA (USA)
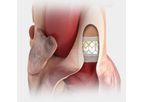
Edwards Lifesciences is the global leader in the science of heart valves and hemodynamic monitoring. Driven by a passion to help patients, the company partners with clinicians to develop innovative technologies in the areas of structural heart ...
The first transcatheter heart valve approved for pre-stented transannular patches. Also approved for use in the pulmonic position in conduits and ...

based inAbbott Park, ILLINOIS (USA)
We create breakthrough products – in diagnostics, medical devices, nutrition and branded generic pharmaceuticals – that help you, your family and your community lead healthier lives, full of unlimited possibilities. Today, 109,000 of us are working ...

based inBrea, CALIFORNIA (USA)
Established in 1997, Life Science Outsourcing, Inc. was founded to offer contract manufacturing services to emerging medical device companies. As an FDA-registered and ISO 13485-certified Contract Manufacturing Organization, we focus on assembly, ...
Life Science Outsourcing offers turnkey medical device manufacturing and assembly services that can be combined with our engineering and process development solutions to create a streamlined and comprehensive process. ...

based inJiaxing City, CHINA
We, Best Hope Precision Device Co., Ltd. are engaged in design and manufacture precision injection molds, typically with rich experience for medical devices. A Leading Manufacturer of Molds and Injection Molded Parts for Medical Devices. In past ten ...
IV Infusion sets are used in devices that provide the proper volume of intravenous medicines, fluids etc. delivered at a certain flow rate. Basically, an IV Infusion set consists of long extruded tube, Connectors, Drip Chamber, Spike and Controller ...

based inScottsdale, ARIZONA (USA)
Confluent Medical Technologies is dedicated to working collaboratively with our customers, taking their projects from rapid prototype into high volume production. Our unparalleled technical expertise, proven experience and partnership with our ...

based inRedwood City, CALIFORNIA (USA)
A global leader in interventional pulmonology, planning tools, and treatments for obstructive lung disease. Based in Redwood City, California, and Neuchâtel, Switzerland, Pulmonx is the maker of the Zephyr Endobronchial Valve. The Zephyr® Valve was ...
Sizing and Placement in One Step: Easy to use. Quick and accurate sizing of the length and width of the airways. Equivalent catheter sizes to valve sizes. Available in a “J” configuration to allow the bronchoscope access to more ...

based inMelville, NEW YORK (USA)
FONAR was incorporated in 1978, making it the first, oldest and most experienced MR manufacturer in the industry. FONAR introduced the world's first commercial MRI (a whole-body MRI scanner) in 1980, and went public in 1981. In 1982, FONAR ...