- Home
- Companies
- marshall islands
- in vitro diagnostic use
Refine by
In Vitro Diagnostic Use Suppliers Serving Marshall Islands
49 companies found

based inYork, UNITED KINGDOM
Abingdon Health is a world leading developer and manufacturer of high-quality rapid lateral flow tests across all industry sectors, including healthcare, animal health and plant health. We take projects from initial concept through to routine and ...

based inPhiladelphia, PENNSYLVANIA (USA)
With extensive experience optimising processes for analytical, production and R&D laboratories across multiple industries and regulatory environments, we provide consistent, quality informatics solutions. We operate on a truly global basis, serving ...

based inIrvine, CALIFORNIA (USA)
MP Biomedicals is a worldwide corporation developing, manufacturing and distributing products for the life science, in vitro diagnostics (IVD), fine chemicals and dosimetry markets. Through our global operational network, we provide research and ...
The Neonatal 17OHP ELISA Kit is intended for the measurement of 17-alpha hydroxyprogesterone (17OHP) in a whole blood ...

based inKAARINA, FINLAND
Labmaster Oy Ltd. is a company engaged in the design, development, and manufacturing of various products, ensuring they meet specified criteria through proper storage, usage, and transportation guidelines. They emphasize adherence to instructions ...
The Labmaster LUCIA™ MxA Kit is a sophisticated in vitro diagnostic tool used for the quantitative assessment of the Myxovirus resistance protein A (MxA) in whole blood samples. This ...

based inUpper Coomera, AUSTRALIA
Bio Molecular Systems (BMS) is a biotechnology company that designs, manufactures and distributes instrumentation for the life sciences. Established in 2010 and comprising of the founders and senior personnel from the former Corbett Life Sciences ...
The world’s first magnetic induction cycler is now a registered medical device with CE-IVD and TGA approval. Mic IVD is manufactured under an ISO 13485:2016 Quality Management System. ...

based inHellerup, DENMARK
BioPorto offers high-quality monoclonal antibodies for pharmaceutical research and IVD testing in areas such as diabetes, obesity, and allergy. BioPorto is an in vitro diagnostics company focused on saving lives and improving the quality of life ...

based inUdine, ITALY
Ulisse BioMed is an Italian healthcare biotech company with corporate offices in Udine, Italy and with R&D and Manufacturing labs in Trieste, Italy. Ulisse BioMed is creating novel, innovative and transformative diagnostic platforms and products. ...
The assay is intended for the qualitative detection of RNA from SARS-CoV-2 in clinician-collected nasopharyngeal swab specimens from patients with signs and symptoms suggestive of COVID-19 (e.g., fever and/or symptoms of acute respiratory ...

based inNanjing, CHINA
Founded in 2002, Jiji Biotech Co., Ltd.is a medical and health industry chain that integrates independent research and development, large-scale production and professional marketing. It has many subsidiaries and offices at home and abroad. the ...
Can be used as a aid in identifying individuals with an adaptive immune response to SARS-CoV-2, indicating recent or prior infection, it should not be used to diagnose acute SARS-CoV-2 infection. ...

based inSomerset, NEW JERSEY (USA)
Even the most sophisticated, cutting-edge medical diagnostics aren’t of value unless we can get them to those who need them most. At Access Bio, we believe every life is precious, and every human on earth deserves the opportunity to live a healthy ...
The CareStart™ COVID-19 MDx RT-PCR is a real-time reverse transcription polymerase chain reaction (RT-PCR) test intended for the qualitative detection of nucleic acid from SARS-CoV-2 in respiratory specimens (such as nasopharyngeal, ...

based inCoralville, IOWA (USA)
A champion of life sciences research for more than 30 years, IDT develops and manufactures nucleic acid products that support the life sciences industry. Areas of focus include academic and commercial research, agriculture, medical diagnostics, ...
We have combined our proven oligo manufacturing expertise and ISO 13485 certified production processes to deliver GMP-manufactured MGB Eclipse Probes and companion primers. Together, these probe and primer pairs are ideal for genotyping qPCR assays. ...

based inWhite City, OREGON (USA)
Biomed Diagnostics is the resulting collaboration of two physicians who found themselves vulnerable without the necessary equipment during a mercy mission in Central America. Without the necessary refrigeration, practical supplies and laboratory ...
Biomed Diagnostics' VTM-C19 Transit Tube is intended for on-site collection and transport to the testing laboratory of human clinical specimens containing SARS-CoV-2 — the virus that causes COVID-19 disease in humans and/or (USA only) ...

based inPoway, CALIFORNIA (USA)
Diazyme uses its proprietary enzyme technologies to develop diagnostic reagents which can be used on most automated chemistry analyzers in user-friendly formats. Diazyme's products include test kits for diagnosis of cardiovascular disease, liver ...

based inGuangzhou, CHINA
Guangzhou Wondfo Biotech Co., Ltd., headquartered in Guangzhou science city, has been focusing on the R&D, production, sales and service of point-of-care testing (POCT) products and providing customers with professional rapid diagnosis and chronic ...
RBD (receptor binding domain) of 2019-nCoV virus S1 protein is the region that binds to the ACE2 receptor on human cells as the first step of virus invading. The majority and most potency neutralizing antibody targets the epitopes on RBD, therefore ...

based inQuilmes,, ARGENTINA
Diconex SA is a company dedicated to design, develop, produce and market clinical chemistry analyzers, used mainly by hospitals, clinics, veterinary clinics and laboratories, scientific research center, human laboratories, wine and food ...
The Clinical Chemistry Autoanalyzer InCCA Counter 18sc is a measuring instrument and characterization of blood cells for in vitro diagnostic use, meassure 18 of the most important blood parameters. ...

based inNewtown, AUSTRALIA
Genetic Signatures was founded in 2001 by the late Dr. Geoffrey Grigg. We are the developers of 3base technology which is the cornerstone of our EasyScreen Pathogen Detection Kits. Our proprietary technology provides hospital and pathology ...
The STI Genital Pathogen and Reproductive Health Reagents are rapid in vitro nucleic acid amplification assays for the qualitative detection of sexually transmitted pathogens as well as commensal and reproductive health-associated ...

based inPoway, CALIFORNIA (USA)
Located in San Diego County, California, an area well known for biotechnology and scientific discovery, CTK Biotech develops and manufactures innovative immunodiagnostic tools and point of care diagnostic test kits for the IVD community worldwide. ...
The RaFIA Immunofluorescence Analyzer is a fluorescence immunoassay analyzing instrument intended for use by healthcare professionals to aid in the diagnosis of conditions such as cardiovascular disease, pregnancy, infection, diabetes, renal injury ...

based inHayward, CALIFORNIA (USA)
Novodiax is dedicated to advancing tissue-based diagnostics and immunoassays through innovation. Our signature technology platform is a proprietary polymer-based detection system. Based on this technology we have developed a series of highly ...
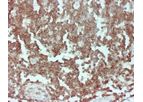
Cluster of Differentiation 45 (CD45) pHRP Anti-Human Staining Kit immunohistochemistry (IHC) reagent is for use on frozen and formalin-fixed-paraffin-embedded tissues. The CD45 antigen is a protein which was originally called Leukocyte Common ...

based inSan Diego, CA 92126 USA, CALIFORNIA (USA)
America Diagnostics, Inc. is a California company that develops and produces IVD devices and diagnostics raw materials. The quality of AmeriDx products is guaranteed by its quality management system. All the products are manufactured in a clean ...
This product is for in vitro diagnostic use, following guidance from the FDA for Emergency Use Authorizations of tests submitted for approval on March 16, 2020. This test has not ...

based inSan Diego, CALIFORNIA (USA)
Epitope Diagnostics Inc., is a certified medical device manufacture company that focuses on niche immunoassays for the global diagnostics and research industries. We specialize in ELISA kits, rapid tests, and antibodies. Epitope Diagnostics Inc. ...
This ELISA (enzyme-linked immunosorbent assay) kit is intended for the qualitative detection of Helicobacter pylori antigen in feces. This assay is a useful tool in the diagnosis of active H. pylori infections. This ELISA kit is for ...

based inHouston, TEXAS (USA)
Azure Biotech Inc is a global biotechnology company that specializes in research & development, as well as the production and sales of diagnostic reagents, POCT analyzers and biological materials. Azure Biotech is also heavily involved in research ...
Webox Cube Type Reader is researched and developed through the modern photoelectric technology, which provide quantitative analysis for colloidal gold reagent strips. With the use of various types of colloidal gold reagents, the relevant ...