Refine by
Clinical Linked Data Articles & Analysis
468 news found
The U.S. House Appropriations Committee’s Fiscal Year 2027 Military Construction and Veterans Affairs (MilCon/VA) appropriations report highlights the role of regenerative wound technologies, including fish-skin-derived products, in limb preservation and value-driven care within the Veterans Health Administration (VHA).The report acknowledges the ongoing burden of chronic, complex, and ...
ByKerecis
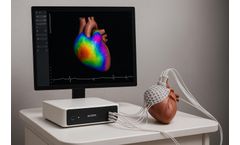
Corify Care today announced a clinical milestone with two studies published in Heart Rhythm and Europace, validating its 4-D global cardiac mapping technology for atrial and ventricular arrhythmias in a single beat.Compared with conventional mapping, which often requires rhythm induction or dependence on pre-procedural imaging, the ACORYS mapping system captures global cardiac electrical activity ...
MADRID, Spain – April, 2026 — Corify Care today announced that the U.S. Food and Drug Administration (FDA) has granted clearance for the ACORYS® System, the first imageless, non-invasive cardiac mapping platform capable of delivering a global, four-chamber view of cardiac electrical activity in real time.The milestone represents a fundamental shift in electrophysiology by combining ...
Orlando, FL – April 16, 2026 – Kerecis, a developer of fish-skin-based tissue regeneration products, reported new clinical data at the American Burn Association Annual Meeting indicating that intact fish-skin grafts reduce hospital length of stay for severely burned patients requiring skin grafting.The study is retrospective and propensity-matched, examining ...
ByKerecis
Charlotte, North Carolina – April 7, 2026. Kerecis will highlight its intact fish-skin platform and new clinical data at SAWC Spring 2026, taking place April 8–11 at the Charlotte Convention Center. The company will showcase intact fish-skin grafts alongside Biatain wound dressings and discuss expanded multi-specialty adoption. Booth presence is at Booth 1508; clinical insights and trial results ...
ByKerecis
BC Platforms today announced enhancements to BC Mosaic, its AI-powered trusted research environment (TRE), now available on AWS Marketplace. The update delivers an all-new user interface and enables secure, compliant analysis of complex clinical, real-world, and multi-omics data within a single TRE, simplifying deployment, configuration, and lifecycle management.BC Mosaic is designed to ...
Hemostemix Inc. (“Hemostemix” or the “Company”) (TSXV: HEM, OTCQB: HMTXF; FSE:2VF0), the leading autologous (patient’s own) stem cell therapy company treating no-option patients in Florida suffering in pain from Chronic Limb Threatening Ischemia, Peripheral Arterial Disease and Angina, is pleased to announce its webinar topic for Thursday, August 21, 2025. Croom ...
An Abbreviated New Drug Application (ANDA) is an application submitted to the U.S. FDA to demonstrate that a generic drug is equivalent to a previously approved Reference Listed Drug (RLD) in terms of safety, efficacy, and quality. The ANDA contains information used for the review and approval of a generic drug product, and does not typically require preclinical and clinical trial data, but ...
SOPHiA GENETICS (Nasdaq: SOPH), a cloud-native healthcare technology company and a global leader in data-driven medicine, today launched the new generation of the SOPHiA DDM™ Platform. The new-gen SOPHiA DDM™ Platform offers a new architecture designed to more efficiently compute large amounts of data at-scale, while elevating customer experience with integrated access to multimodal ...
CanaQuest Medical Corp (“CanaQuest” or the “Company”) (OTC: CANQF), is pleased to announce strategic additions to its executive team with expertise in conducting clinical trials and US FDA drug approval success. The team plans to effectively navigate the regulatory approval process for Drug Candidate, CQ-001 (cannabidiol + proprietary API composition), to treat rare ...
Tempus, a leader in artificial intelligence and precision medicine, today announces an expansion of its collaboration with AstraZeneca, which leverages Tempus Next to arm physicians with technology that supports the delivery of guideline-directed biomarker testing for patients with non-small cell lung cancer (NSCLC). The magnitude and frequency of updates to oncology guidelines each year make it ...
ByTempus
Introduction Intraoperative irrigation is a commonly used method aimed at reducing microbial burden by removing tissue debris, metabolic waste, and tissue exudate from the surgical field prior to site closure.1 With the absence of universally established standard of care and robust clinical data, the selection of appropriate irrigation solution remains a topic of debate. Isotonic saline has ...
Caresyntax, the leading vendor of enterprise-scale data-driven surgical intelligence solutions to make surgery safer and smarter, today announced its highlights and achievements of 2023, as the company has continued its strong growth journey. Dennis Kogan, co-founder, and CEO: “Caresyntax celebrated its 10-year anniversary in 2023 and accelerated our journey of innovation and expansion ...
Caresyntax, the leading vendor-neutral surgical data and intelligence platform converging AI-powered software, devices, and clinical services, today announced the launch of Caresyntax Clinical Data as a Service (CDaaS) for Medtech industry partners. ...
The collaboration will leverage a secure infrastructure that is compliant with data privacy, information security, and regulatory concerns across the geographies. ...
Caresyntax, the leading vendor-neutral enterprise surgical data and intelligence platform designed to make surgery smarter and safer by converging AI-powered software, devices, and clinical services, today announced a partnership with ARC (Accelerate, Redesign, Collaborate) Sheba’s Global Innovation platform to establish and replicate the ARC Surgical ...
With more than 60 years’ expertise in respiratory diagnostics, Vitalograph is the only company in the world specialized in delivering clinical trial services exclusively for pulmonary function-focused studies. Speaking about the recent additions to their portfolio of services, Richard James, Executive Vice President and Chief Operating Officer of Vitalograph Clinical Trials says: ...
Intermountain Primary Children’s Hospital First in the Country to Implant this Next Generation FDA-Cleared Device Company Welcomes Vice President of Sales & Marketing, Mark Geiger, Who Lives with Hydrocephalus Anuncia Medical Inc. (“Anuncia” or the “Company”), a Company specializing in advancing innovations for implantable and external cerebrospinal fluid ...
New Subgroup and further analyses from PULSAR, PHOTON and CANDELA provide insights into durability results of extended treatment intervals, patient characteristics as well as efficacy and safety of intravitreal aflibercept 8 mg In total, 18 presentations on aflibercept 8 mg and Eylea (aflibercept 2 mg) reinforce Bayer’s commitment to advancing treatment of patients with exudative retinal ...
ByBayer AG
Daxor Corporation (NYSE: DXR), the global leader in blood volume measurement technology, today announces new data validating the benefits of the Company’s BVA-100 diagnostic in optimizing individualized therapy for heart failure patients. Data were presented at the Technology Heart Failure Therapeutics Conference, which focuses on device and technology-based treatments in heart failure. ...