Refine by
Clinical Program Articles & Analysis
413 news found
Bremen, Germany – May 4, 2026 — Kerecis, the company pioneering the use of sustainably sourced intact fish skin in cellular therapy and tissue regeneration, will play a prominent scientific and educational role at EWMA-DEWU 2026 Conference, taking place May 6–8 at the Messe & Congress Centrum Bremen in Bremen, Germany.This year EWMA, The European Wound Management Association meeting, is held in ...
ByKerecis
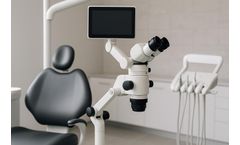
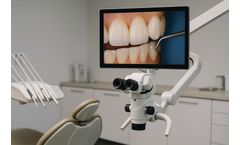
ImageWorks announced a new partnership with the MOD Institute to integrate the Mielscope digital microscope into MOD’s hands-on training programs and clinical environment. The collaboration aims to provide MOD clinicians and students unprecedented visibility into complex dental techniques while supporting modern ergonomic workflows.“After using the Mielscope in ...
ImageWorks announced a strategic partnership with The MOD Institute to integrate the Mielscope digital microscope into MOD Institute's hands-on dental training programs and clinical environment. The collaboration will provide MOD clinicians and students with enhanced visibility into complex techniques while supporting ergonomic workflows in real-world ...
A Unique Challenge and Opportunity for Higher Education Across the country, colleges and universities were forced to make difficult decisions about the Fall 2020 semester as a result of the ongoing COVID-19 pandemic. Rollins College was no exception. However, early in their planning, administrators saw a unique opportunity to preserve some of the traditional college experience by utilizing new, ...
CanaQuest Medical Corp (“CanaQuest” or the “Company”) (OTC: CANQF), is pleased to announce strategic additions to its executive team with expertise in conducting clinical trials and US FDA drug approval success. The team plans to effectively navigate the regulatory approval process for Drug Candidate, CQ-001 (cannabidiol + proprietary API composition), to treat rare ...
The OCEANIC-AF (atrial fibrillation) study with asundexian is part of the Phase III OCEANIC clinical trial program enrolling more than 27,000 patients in over 40 countries Asundexian is being evaluated as a potential improved treatment option in stroke prevention and could be part of an entirely new class of treatment options in thrombosis management that aims ...
ByBayer AG
Lesnick is a scientifically driven executive with deep expertise managing global research and development programs from pre-clinical stage through Phase 3, as well as regulatory affairs and quality assurance. ...
The following new abstracts for aflibercept 8 mg will be presented at ARVO 2023: The pooled safety analysis compares the safety of aflibercept 8 mg with aflibercept 2 mg across the clinical trial program which includes data from the phase II trial CANDELA in nAMD and the pivotal phase II/III trial PHOTON in DME and the phase III trial PULSAR in nAMD. ...
ByBayer AG
Bayer presents first Phase 1 results from its most advanced clinical Immuno-Oncology program, aryl hydrocarbon receptor (AhR) inhibitor BAY2416964 New preclinical data on Bayer’s DGK zeta inhibitor being featured in oral presentation in New Drugs on the Horizon session New preclinical data on mutEGFR HER2 program, currently in Phase 1, ...
ByBayer AG
Phase III study ARASTEP initiated in hormone-sensitive prostate cancer, in patients with high-risk biochemical recurrence (BCR) who have no evidence of metastatic disease by conventional imaging and a positive PSMA PET CT at baseline Fifth major clinical trial for androgen receptor inhibitor (ARi) darolutamide, covering prostate cancer from early to metastatic stage Bayer ...
ByBayer AG
According to Irene Ghobrial, MD, Director of the Clinical Investigator Research Program at Dana-Farber Cancer Institute, Boston, and senior author of the study “this new data represents another great milestone, both for our understanding of MM and abilities to prevent disease progression”. ...
The company’s founding team includes highly experienced executives and internationally renowned experts with track records in developing retinal gene therapy programs from discovery to clinical stage: Dr. Caroline Man Xu (Co-founder and CEO), Prof. ...
Efficient transfer of larger gene via ViGeneron’s proprietary technology platforms REVeRT and vgAAV ViGeneron advances its preclinical programs for Stargardt disease and Retinitis Pigmentosa into IND-enabling activities and clinical stage development ViGeneron GmbH, a next-generation gene therapy company, today announced the presentation of ...
The Company expects the protocols for its clinical program to be influenced by the preclinical data and the Phase 1 clinical trial experience. ...
The company is already advancing multiple novel biology, first-in-class programs towards the clinic and has more than a dozen similar pipeline opportunities emerging in early discovery. ...
ByBayer AG
“We have demonstrated preclinical safety and efficacy of our oral, small molecule program through well designed pre-clinical studies, and are now poised to take a major step into clinical development. ...
“As an oral small molecule, our lead program is a highly differentiated approach that targets tau self-association, the beginning stages of the tau aggregation cascade, for the treatment of neurodegenerative diseases. ...
The financing will be used to advance NextPoint’s two lead precision immuno-oncology programs into the clinic, both targeting the newly discovered HHLA2 pathway to activate anti-tumor immune responses. ...
ByBayer AG
In parallel, the ongoing Phase III clinical development programs (ARANOTE and ARASEC) with darolutamide aim to broaden its use even further by offering treatment options in the metastatic hormone-sensitive setting without concomitant chemotherapies. ...
ByBayer AG
The company provides small molecule drug discovery services and its comprehensive enabling technology platforms, coupled with roots to Nobel Prize winning science and the Max Planck Institute of Biochemistry, have enabled prominent contributions to several lead optimization programs and clinical-stage compounds. Proteros’ scientific rigor can accelerate ...