Copd Patient Articles & Analysis
26 news found
Chronic Obstructive Pulmonary Disease (COPD) is a global health burden characterized by persistent airflow limitation and progressive lung damage. Despite extensive research, translating findings from bench to bedside remains a challenge. Precision Cut Lung Slices (PCLS) have emerged as a versatile tool in COPD research, offering unique advantages for understanding disease mechanisms and ...
It can aid in the diagnosis of asthma and COPD and can also help identify patients who are at risk of progression to COPD. [1] The tremoflo® C-100 assesses respiratory function through simple tidal breathing and can be a valuable tool when compiling a detailed picture of respiratory function. It is particularly useful in enabling the very ...
” Bill Heiden, Third Pole’s Chairman added, “This strategic investment in Third Pole Therapeutics is directly aligned with both organization’s mission to advance unique, disruptive, cost-effective solutions that improve outcomes in heart and lung-failure patients. It also provides validation that Third Pole’s technology represents the best in ...
Having a treatment that can help patients breathe easier and live a better quality of life, without surgical intervention, will be a major step forward in COPD care in Japan.” Over 600,000 Japanese patients suffer from severe COPD, including approximately 100,000 patients with severe disease where ...
Pulmonx’s Zephyr® Endobronchial Valve, Chartis® Pulmonary Assessment System and StratX® Lung Analysis Platform are designed to assess and treat patients with severe emphysema/COPD who despite medical management are still profoundly symptomatic. ...
(NASDAQ: XAIR) a medical device and biopharmaceutical company focused on developing inhaled nitric oxide (NO) for the treatment of patients with respiratory conditions, including serious lung infections and pulmonary hypertension, and, through its affiliate Beyond Cancer, Ltd., ultra-high concentration nitric oxide (UNO) for the treatment of solid tumors, today announced ...
Pulmonx’s Zephyr® Endobronchial Valve, Chartis® Pulmonary Assessment System and StratX® Lung Analysis Platform are designed to assess and treat patients with severe emphysema/COPD who despite medical management are still profoundly symptomatic. ...
The AeriSeal System is used to close collateral air channels in a target lung lobe of a patient with severe COPD/emphysema, making the patient eligible to then undergo Bronchoscopic Lung Volume Reduction (BLVR) with Zephyr Valves. ...
Pulmonx’s Zephyr® Endobronchial Valve, Chartis® Pulmonary Assessment System and StratX® Lung Analysis Platform are designed to assess and treat patients with severe emphysema/COPD who despite medical management are still profoundly symptomatic. ...
Pulmonx’s Zephyr® Endobronchial Valve, Chartis® Pulmonary Assessment System and StratX® Lung Analysis Platform are designed to assess and treat patients with severe emphysema/COPD who despite medical management are still profoundly symptomatic. ...
eNOfit™, Third Pole’s novel nitric oxide mobile-wearable device designed to provide “on-the-go” treatment for patients suffering from severe COPD and ILD, to enter clinical trials in the second half of this year Funding will support device optimization and clinical trials; Pivotal trial data expected in the second half of 2023 Third ...
According to WHO, the demonstration of irreversible airway obstruction is essential for diagnosing chronic obstructive pulmonary disease (COPD), which is the third leading cause of death worldwide. It is estimated that up to 67–81% of patients with COPD remain undiagnosed. Mobile phone-linked spirometers can be used almost anywhere – ...
ByAioCare
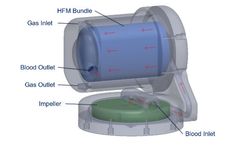
ALung has initiated commercial development of the next generation Hemolung RAS to provide clinicians with the flexibility to support patients across the full spectrum of acute and acute-on-chronic respiratory failure using a single integrated device. ...
The ASCENT study was conducted in patients with moderate-to-very severe chronic obstructive pulmonary disease (COPD) and cardiovascular disease and / or significant cardiovascular risk factors. This patient population is of particular relevance as cardiovascular disease is the most common and significant comorbidity of COPD, with ...
The ASCENT study was conducted in patients with moderate-to-very severe chronic obstructive pulmonary disease (COPD) and cardiovascular disease and / or significant cardiovascular risk factors. This patient population is of particular relevance as cardiovascular disease is the most common and significant comorbidity of COPD, with ...
Steve Harris, Circassia’s Chief Executive, said: “We are delighted with the FDA approval of Duaklir®, which we believe will provide a valuable treatment option for the significant number of patients with COPD in the United States. The addition of Duaklir® to our portfolio further strengthens our range of marketed respiratory products and we ...
Tudorza®’s expanded label now includes data from the phase IV ASCENT study, which was conducted in patients with moderate to very severe COPD and cardiovascular disease and / or significant cardiovascular risk factors. The study demonstrated that Tudorza® is effective at reducing COPD exacerbations with no increase in major ...
Tudorza®’s expanded label now includes data from the phase IV ASCENT study, which was conducted in patients with moderate to very severe COPD and cardiovascular disease and / or significant cardiovascular risk factors. ...
We believe that based on its broad clinical database, Duaklir®, if approved, has the potential to become an important new treatment option for COPD patients in the United States. In addition, the inclusion of clinical data in Tudorza®’s prescribing information demonstrating cardiovascular safety and reductions in COPD ...
We believe that Duaklir®, if approved, has the potential to provide an important treatment option for the significant number of patients with COPD in the United States. In addition, the positive cardiovascular safety and COPD exacerbation reduction results achieved in the Tudorza® ASCENT study offer the potential to include unique new ...