Refine by
Medical Evaluation Articles & Analysis
36 news found
As a leading provider of product advisory and compliance services solutions, Proregulations understands the complexity and importance of 510(k) applications and supports medical device manufacturers worldwide from initial evaluation to final marketing. ...
STEMart, a US-based provider of comprehensive services for all phases of medical device development, has announced the launch of its new Accelerated Aging Testing Services for medical device manufacturers to ensure the long-term reliability and safety of their products. ...
BySTEMart
This proactive approach significantly reduces the risk of adverse reactions, ensuring that veterinary drugs are both safe and effective. Evaluating Generic Veterinary Drugs In addition to innovating new drugs, CD Formulation plays a vital role in the evaluation of generic veterinary drugs. Consistency in drug performance and quality is paramount for generics to ...
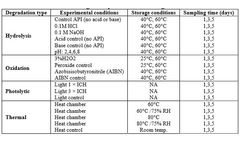
STEMart, a US-based provider of comprehensive services for all phases of medical device development, announced the launch of its new Force Degradation Services to help pharmaceutical and medical device companies evaluate the stability of their drug candidates and finished products under a variety of stress conditions, ensuring their safety and ...
BySTEMart
In these cases, you can turn to medical aesthetic technologies. All the technologies that treat muscle toning work on adipose tissue, which can accumulate in different places: in the inner thigh, under the arms, on the buttocks, etc. Biotec Italia Medical, however, has re-evaluated this concept, going to work directly on the muscle, which makes ...
Department of Defense (DoD) Congressionally Directed Medical Research Programs (CDMRP) to evaluate efficacy of the company’s single-dose chikungunya virus virus-like particle (CHIKV VLP) vaccine candidate. Emergent will begin the planning phase, the first of two phases, in collaboration with the Armed Forces Research Institute of Medical ...
iRhythm is included in the Medtech Big 100, a ranking of the world’s largest medical device companies, for the second year in a row. Medical Design & Outsourcing says it evaluates companies for the list based on annual revenue, research and development spending, number of employees, key leaders, headquarters locations, and company ...
These findings together with the now published immunological results support the rationale of the ASSET innovation milieu coordinated by Diamyd Medical where precision prevention strategies will be evaluated using the diabetes vaccine Diamyd®. ...
Inari Medical, Inc. (NASDAQ: NARI) (“Inari”) a medical device company with a mission to treat and transform the lives of patients suffering from venous and other diseases, announced planned enrollment of the DEFIANCE trial. DEFIANCE is a randomized controlled trial (“RCT”) comparing the clinical outcomes of patients with iliofemoral deep vein thrombosis ...
We could not have achieved this without the help of following departments and staff: Brendan McBrine and Iris Wen (CCRO) Kathleen Charbonnier, Kathy Oconnor and Jenn Lilly (Nursing) Christine Finn (Pharmacy) Lisa Bove, Kathleen Melanson and Delia Barry (Clinical Lab) Medical Evaluation Unit We are optimistic that this may become the standard of care for ...
“These results are very encouraging and the next step is together with collaborators and authorities evaluate the regulatory requirements to get the diabetes vaccine Diamyd approved as a disease-modifying therapy in individuals diagnosed with LADA”, says Ulf Hannelius, CEO of Diamyd Medical. ...
STEMart, a provider of integrated medical device CRO services dedicated to medical device and diagnostic clinical development, expands its Biocompatibility Testing services and introduces biocompatibility evaluation of breathing gas pathways in medical devices. ...
BySTEMart
TORONTO--(BUSINESS WIRE)-- Titan Medical Inc. (Nasdaq: TMDI; TSX: TMD), a medical device company focused on the development and commercialization of innovative surgical technologies for single access robotic-assisted surgery (RAS), today announced financial results for the three months ended March 31, 2022. ...
NeuroOne Medical Technologies Corporation (NASDAQ: NMTC) ("NeuroOne" or the "Company"), a medical technology company focused on improving surgical care options and outcomes for patients suffering from neurological disorders, today announces its operating results for the second quarter of fiscal year 2022 ended March 31, 2022. ...
STEMart, a provider of integrated medical device CRO services dedicated to medical device and diagnostic clinical development, now launches Pyrogenicity Testing service for the medical device industry. ...
BySTEMart
The new $1.2M grant will support a 40-patient clinical trial at the University of Maryland Medical Center (UMMC) evaluating safety and cost-effectiveness characteristics of a novel medical procedure known as percutaneous ultrasound gastrostomy (PUG), which is enabled by CoapTech’s PUMA-G device. ...
ByCoapTech
Food and Drug Administration (FDA) Investigational Device Exemption (IDE) study, to evaluate the safety and efficacy of P-15L Bone Graft for use in transforaminal lumbar interbody fusion (TLIF) surgery in patients with degenerative disc disease (DDD). ...
BioXtreme is proud to announce that a new commercial evaluation agreement was signed with STEPS Budapest Center for Robotic Rehabilitation. The agreement includes a 6- months evaluation period for deXtreme, by the end of which STEPS commits to purchasing the device (pending evaluation criteria are met). ...
Verification is critical for medical device testing, which is why we recommend reading part one if you haven’t already. ...
(Nasdaq: NSPR), a global developer of the CGuard™ Embolic Prevention Stent System (EPS) device for the treatment of Carotid Artery Disease (CAD) and stroke prevention, today announced that its CGuard EPS stent system has received a positive opinion from the National Commission for the Evaluation of Medical Devices and Health Technologies (CNEDIMTS) of the ...