Refine by
Pharmaceutical Medical Articles & Analysis
185 news found
Tubes Pvt Ltd, a leading supplier of specialty chemicals in India, today announced the expansion of its product line to better serve the rising demand in pharmaceutical, medical device, and industrial manufacturing sectors. The company’s latest initiatives are aligned with India’s "Make in India" vision, aiming to deliver advanced chemical solutions that meet ...
Obtaining the Marketing Authorisation Application (MAA) is a key step in the commercialization of drugs in the EU, and also an important help to promote the global market expansion of pharmaceutical companies. The MAA is complicated by the need to consider both European economic integration and the independence of individual member states. ...
As a form of DMF in the EU, the ASMF is used to support MAA or MAV by manufacturers of pharmaceutical preparations for human or veterinary use. The ASMF applies to applications for new active substances, pharmacopeial active substances (included in the EP) and existing active substances (excluded from the EP). ...
U.S. medical device regulations require manufacturers of most class II and a small number of class I devices to file a 510(k) unless they qualify for an exemption. 510(k) is a premarket technical document submitted by the manufacturer to the FDA before the medical device product enters the US market to prove that the product has the same safety and effectiveness ...
Our service areas range from chemicals, pharmaceuticals, medical devices, cosmetics, food & dietary supplements to agrochemicals & biocides. Our featured services include but are not limited to FDA Medical Device Registration, 510(k) Premarket Notification, Drug Master File Document, FDA ANDA, FDA NDA, IND Application, New Dietary ...
Our service areas range from chemicals, pharmaceuticals, medical devices, cosmetics, food & dietary supplements to agrochemicals & biocides. Our featured services include but are not limited to FDA Medical Device Registration, 510(k) Premarket Notification, Drug Master File Document, FDA ANDA, FDA NDA, IND Application, New Dietary ...
By facilitating the development of cutting-edge therapies, our products support advancements in various medical fields, including gene therapy, vaccine creation, and oncology treatments. “At Biopharma PEG, we are dedicated to providing innovative, high-performance PEG products tailored for the pharmaceutical and biotech sectors,” stated Brenda, ...
Biopharma PEG, a leader in PEG derivatives, is excited to announce the expansion of its high-purity Multi-Arm PEG linker product line, catering to the evolving needs of the medical and bioorganic fields. These advanced PEG linkers are available in various functional groups and molecular weights ranging from 1k to 40k, offering unmatched versatility and performance for research ...
CanaQuest Medical Corp (“CanaQuest” or the “Company”) (OTC: CANQF), is pleased to announce strategic additions to its executive team with expertise in conducting clinical trials and US FDA drug approval success. The team plans to effectively navigate the regulatory approval process for Drug Candidate, CQ-001 (cannabidiol + proprietary API composition), to treat rare ...
ABOUT XTALKS Xtalks, powered by Honeycomb Worldwide Inc., is a leading provider of educational webinars and digital content to the global life science, food, healthcare and medical device communities. Every year, thousands of industry practitioners (from pharmaceutical, biotechnology, food, healthcare and medical device companies, private & ...
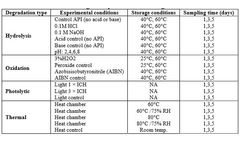
STEMart, a US-based provider of comprehensive services for all phases of medical device development, announced the launch of its new Force Degradation Services to help pharmaceutical and medical device companies evaluate the stability of their drug candidates and finished products under a variety of stress conditions, ensuring their safety and ...
BySTEMart
About Xtalks Xtalks, powered by Honeycomb Worldwide Inc., is a leading provider of educational webinars and digital content to the global life science, food, healthcare and medical device communities. Every year, thousands of industry practitioners (from pharmaceutical, biotechnology, food, healthcare and medical device companies, private & ...
The multi-target research and development partnership will focus on severe skin diseases The collaboration leverages etherna’s proprietary mRNA and lipid nanoparticle (LNP) technology with Almirall’s expertise in the dermatology space In addition to upfront and technology access payments, etherna is eligible to receive €300 million in development and commercial ...
ByeTheRNA
After a procedure, the Caresyntax platform provides insights that help surgeons benchmark and improve their care, hospital administrators use surgical resources more efficiently, medical device companies advance better products, and insurance companies understand risk and devise more tailored policies. ...
Amerigo Scientific, as a reliable product and service distributor for research institutions and enterprises in the biological, chemical, pharmaceutical, medical, and other industries, recognizes the increasing need for reliable and ready-to-use solutions that can optimize lab workflows and ensure consistent results. ...
Vizient serves approximately 97% of the nation's Academic Medical Centers, more than 50% of the nation's acute care health system, and serves more than 20% of the nation's ambulatory market. This contract enables TOMI to supply SteraMist systems to a wide range of healthcare providers, including academic medical centers, pediatric facilities, and community health ...
TOMI products are designed to service a broad spectrum of use sites, including, but not limited to, hospitals and medical facilities, biosafety labs, pharmaceutical facilities, commercial and office buildings, schools, restaurants, meat and produce processing facilities, and police and fire departments. ...
Efficient leadership plays a huge role in bringing a clinical trial to fruition. Pharmaceutical companies are not immune to making mistakes and facing failures. ...
The organization aims to supply the people affected in 28 European countries with free medical mouth-nose-protection for their caregivers as a form of external protection. ...
ViGeneron’s pipeline in gene therapy addresses ophthalmic diseases with high unmet medical need, including two programs in development for undisclosed indications where no approved treatment options are currently available. ...