Refine by
Respiratory Device Articles & Analysis
23 news found
Vitalograph has joined forces with MedM Inc. to further enhance patient respiratory monitoring experience with digital health capabilities. The MedM health monitoring app is paired with Vitalograph’s Asthma-1 BT and Lung Monitor BT Smart devices. ...
Breakthrough pulse oximetry (SpO2 ) optical sensor solution offers medical-grade accuracy of oxygen level measurement across skin tones and while in motion Patented SpO2 sensor chipset, integrated processing and reference design capability to power the next generation of medical and consumer wearable devices BioIntelliSense, a continuous health monitoring and clinical intelligence company, ...
Fluidda, leader in the field of Functional Respiratory Imaging and developer of the medical device Broncholab®, today announced an exclusive collaboration with Nanopharm, an Aptar Pharma company and a leader in contract research and development services for orally inhaled and nasal drug products (OINDPs). ...
ByFluidda
(https://intin.kr/en/) has been recently honored with the 'Innovation Award' from CES (Consumer Electronics Show) for its 'Oview Multi Device' product, a respiratory diagnosis and treatment system. CES is the world's biggest & influential tech event, held annually in the USA. ...
John Pritchard, PhD is an independent consultant specializing in strategic approaches to developing respiratory devices, drugs and digital health. In addition, he is a Director for several companies and sits on three Scientific Advisory Boards. ...
It is a respiratory monitoring system used to rapidly assess a patient’s respiratory status. The device is FDA cleared and approved by Health Canada for COVID-19 Emergency Use. About MediPines MediPines Corporation, based in California, is a market leader in respiratory assessment and monitoring of pulmonary gas exchange. ...
Russian regulatory certification for USCOM 1A Cardiac Output Monitor, Russia a significant and growing European market, Uscom continues global expansion. USCOM 1A has received Russian Certification of Registration for sale into the Russian market. The regulatory process is usually 12-18 months but took approximately 3 years following delays with the COVID pandemic. Uscom has worked closely with ...
Moisture in breath samples can adversely impact the accuracy of breath analysis and damage devices. As such, respiratory device providers seek robust solutions that reduce moisture from breath samples and help yield accurate results. ...
Bothell, Washington - On May 29, 2020, the Centers for Medicare and Medicaid Services (CMS) updated its policy for HCPCS code E0467 (Multi-Function Ventilator) removing “same or similar” restrictions and paving the way for increased access to integrated respiratory care for ventilator users regardless of their device billing history. ...
Patent protected wireless lung function testing technology, Asthma, COPD, and post-COVID syndrome, Spirometry market is predicted to double in 12 months, Globalisation initiative – Europe, SE Asia and the US. Uscom notifies the market of notice to approve CE mark for the Uscom digital ultrasonic SpiroSonic AIR spirometer. The CE mark is recognition of legal compliance with health, safety ...
“We are delighted that our technology is proving robust in allowing leading scientists to push the envelope of respiratory science. With experiments in extreme environments such as high altitude and free diving, these scientists are using our device to better understand compromised lung function due to disease processes. ...
Using the MediPines AGM100®, a respiratory medical device,researchers were able to demonstrate that the new breath-based measurement of gas exchange efficiency has very low variability and is highly correlated with established blood-sampling methods. ...
These respiratory diseases require a precise determination of gas exchange to detect and treat these illnesses. ...
Patients wear a small clip on their nose while breathing normally into a disposable mouthpiece that is connected to a monitor. The device can be delivered to Canada in a few days. “This portable device, while doing complex things, is simple to operate,” Dr. ...
Liberate Medical today announced the results of a second randomized controlled pilot trial of its proprietary medical device, the VentFree Respiratory Muscle Stimulator, the only non-invasive and only breath-synchronized neuromuscular electrical stimulation device used to address respiratory muscle atrophy in mechanically ...
The tested hardware will consist of EmbracePlus, a medical device in a smartwatch form-factor, with clinical-grade sensors that measure heart rate, heart rate variability, temperature, respiratory rate, and electrodermal activity. The device unobtrusively monitors a person's vitals, and sends in-app alerts for patterns suggestive of COVID-19 ...
To meet this growing challenge in the face of rising pandemic, MediPines, a respiratory medical device company, has released a scientific paper on respiratory monitoring in long-term care facilities. ...
Thorasys - Thorasys is a small medium-sized enterprise (SME), based in Montreal, Quebec, Canada, which designs, manufactures and markets respiratory medical devices such as the tremoflo®. Thorasys applies state-of-the-art technology in the development of products for the assessment of lung function.in patients with and without disease. ...
MediPines, a respiratory medical device company, was selected a winner in the special COVID-19 edition of the “Make Your Medical Device Pitch for ...
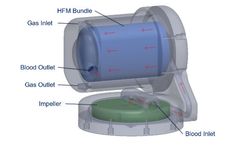
ALung has initiated commercial development of the next generation Hemolung RAS to provide clinicians with the flexibility to support patients across the full spectrum of acute and acute-on-chronic respiratory failure using a single integrated device. The system design will accommodate bedside therapy as well as portability and wearability, further enhancing ...