- Home
- Equipment
- canada alberta
- in vitro diagnostic test
Refine by
In Vitro Diagnostic Test Equipment & Supplies In Canada Alberta
58 equipment items found
Manufactured by:Omega Diagnostics Group PLC based inAlva, UNITED KINGDOM
VISITECT CD4 in-vitro diagnostic test is for use as an aid in the management of patients with pre-diagnosed HIV infection. This visually read test is designed to be used at the point-of-care and therefore has utility in decentralised diagnostic settings. The VISITECT® CD4 Rapid Test is a ...
Manufactured by:Edif Instruments s.r.l. based inRoma, ITALY
The SARS-CoV-2 IgM ELISA kit is an in vitro diagnostic test for the qualitative detection of IgM antibodies to SARS-CoV-2 in human serum. The kit is intended for use as an aid in identifying individuals with an adaptive immune response to SARSCoV-2, indicating recent or prior infection. ...
Manufactured by:PROTIA. iNC, formely known as ProteomeTech Inc. based inSeoul, SOUTH KOREA
PROTIA Allergy-Q 64S panel is an in vitro diagnostic test in the quantitative determination of allergen-specific IgE concentrations in human serum or plasma using immunoblotting technique. Diagnose 63 types of allergens including Total IgE in one ...
Manufactured by:CLONIT srl based inAbbiategrasso, ITALY
DuplicαRealTime Next Factor V H1299R Kit is an in vitro diagnostic test for the detection of the nucleotide substitution A4070G of the blood coagulation Factor V gene, by Real-Time ...
Manufactured by:DxGen Corp. based inGunpo-si, SOUTH KOREA
Test Kit is for in-vitro diagnostic (IVD) use to determine C-reactive protein (CRP) quantitatively in serum/plasma, or whole blood from a human. It provides information for the diagnosis, therapy, and monitoring of inflammatory ...
Manufactured by:PROTIA. iNC, formely known as ProteomeTech Inc. based inSeoul, SOUTH KOREA
PROTIA Allergy-Q 64 Food panel is an in vitro diagnostic test in the quantitative determination of allergen-specific IgE concentrations in human serum or plasma using immunoblotting technique. Diagnose 72 types of food allergens including Total IgE in one ...
Manufactured by:PROTIA. iNC, formely known as ProteomeTech Inc. based inSeoul, SOUTH KOREA
PROTIA Food IgG a is an in vitro diagnostic test with the semi-quantitative determination of food specific IgG (sIgG) in human serum using immunoblotting method. Diagnose 60 types of allergens (inducing food intolerance) in one ...
Manufactured by:PROTIA. iNC, formely known as ProteomeTech Inc. based inSeoul, SOUTH KOREA
PROTIA Allergy-Q 128M panel is an in vitro diagnostic test in the quantitative determination of allergen-specific IgE concentrations in human serum or plasma using immunoblotting technique. Diagnose 118 types of allergens including Total IgE in one ...
Manufactured by:Firalis Group based inHuningue, FRANCE
NEUROLINCS : A non-invasive in-vitro diagnostic test measuring 7494 brain-enriched long non-coding RNAs (lncRNAs) that are specific to neurodegenerative ...
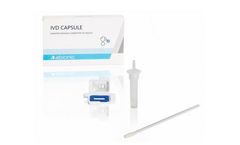
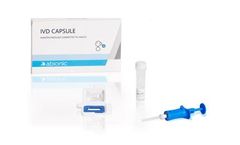
Manufactured by:Abionic SA based inEpalinges, SWITZERLAND
The IVD CAPSULE COVID-19 is a rapid, single-use in vitro diagnostic test that measures the presence of a specific SARS-CoV-2 viral antigen in saliva. Abionic’s COVID-19 saliva test allows for ultra-fast screening of COVID-19 patients at the point-of-care with lab quality ...
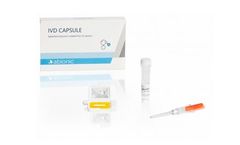
Manufactured by:Abionic SA based inEpalinges, SWITZERLAND
The IVD CAPSULE Ferritin is a rapid, single-use in vitro diagnostic test for the quantitative measurement of ferritin in capillary and venous whole blood, to aid blood diagnostic testing for iron deficiency in ...
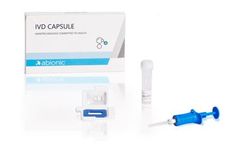
Manufactured by:Abionic SA based inEpalinges, SWITZERLAND
The IVD CAPSULE PSP is a rapid, single-use in vitro diagnostic test for the quantitative measurement of pancreatic stone protein (PSP) in blood. The test is intended to be used in conjunction with other clinical assessments and laboratory findings to aid in the early detection of nosocomial sepsis in ...
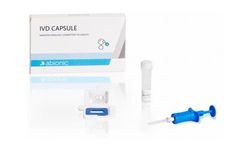
Manufactured by:Abionic SA based inEpalinges, SWITZERLAND
The IVD CAPSULE Aeroallergens is the first rapid, single-use in vitro diagnostic test for the quantitative determination of circulating immunoglobulin E (IgE) specific to the allergen components Phl p 1 + Phl p 5 (combined), Der p 1 + Der p 2 (combined), Alt a 1, Fel d 1 and Can f 1 in uncoagulated human capillary blood collected from the ...
Manufactured by:Abionic SA based inEpalinges, SWITZERLAND
The IVD CAPSULE PSP is a rapid, single-use in vitro diagnostic test for the determination of Pancreatic Stone Protein (PSP) in blood for the calculation of the cSOFA score at the point of care. The cSOFA score is used to assess the severity of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in ...
Manufactured by:CLONIT srl based inAbbiategrasso, ITALY
The PML-RARA t(15;17)(q22;q21) is an in vitro diagnostic test for the detection and identification of the PML-RARA gene fusions resulting from the t(15;17)(q22;q21) , by multiplex one-step Real-Time PCR. The PML-RARA t(15;17)(q22;q21) allows also the detection and quantification of ABL as a endogenous control gene. ...
Manufactured by:CLONIT srl based inAbbiategrasso, ITALY
The AML1-ETO t(8;21)(q22;q22) is an in vitro diagnostic test for the identification and quantification of the AML1-ETO gene fusion transcripts resulting from t(8;21)(q22;q22) , by one-step Real-Time PCR. The AML1-ETO t(8;21)(q22;q22) assay allows also the detection and quantification of ABL as a endogenous control gene. ...
Manufactured by:Coris BioConcept based inGembloux, BELGIUM
In vitro rapid diagnostic test for the detection of SARS-CoV-2 antigen in nasopharyngeal secretions. Since many months, several variants did pop-up from countries where the pandemic was particularly active (i.e. UK, Brazil, India and South Africa) and our test has been proven to detect all of ...
Manufactured by:Labmaster Ltd. based inKAARINA, FINLAND
The Labmaster LUCIA™ CRP Kit is an in vitro diagnostic solution designed to quantitatively measure C-reactive protein (CRP) from whole blood samples. This near-patient diagnostic tool is intended for use with the semi-automated Labmaster LUCIA™ Analyzer, facilitating healthcare professionals in assessing the body's inflammatory ...
Manufactured by:Sysmex Nederland B.V. based inEtten-Leur, NETHERLANDS
The new standard in routine allergy diagnostics. NOVEOS provides patient comfort with only 4 µl sample volume and performs accurate ...
Manufactured by:Infitek Co., Ltd. based inJinan, CHINA
The Cholera Ag O1 Rapid Test is a rapid chromatographic immunoassay for the qualitative detection of Cholera Ag O1 antigen from fecal samples to aid in the diagnosis of O1 group Cholera ...