Refine by
Interbody Implant Equipment & Supplies In Canada
8 equipment items found
Manufactured by:ATEC Spine, Inc based inCarlsbad, CALIFORNIA (USA)
ATEC’s IdentiTi Anterior Lumbar SW Porous Ti Interbody Implants are designed to provide the biological, biomechanical, and imaging characteristics that surgeons seek in a fusion construct. The subtractive process used to manufacture each IdentiTi Implant results in more predictable mechanical performance and enhanced imaging ...
Manufactured by:ATEC Spine, Inc based inCarlsbad, CALIFORNIA (USA)
ATEC’s IdentiTi Cervical Porous Ti Interbody Implants are designed to provide the biological, biomechanical, and imaging characteristics that surgeons seek in a fusion construct. The subtractive process used to manufacture each IdentiTi Implant results in more predictable mechanical performance and enhanced imaging characteristics. IdentiTi ...
Manufactured by:ATEC Spine, Inc based inCarlsbad, CALIFORNIA (USA)
Battalion PC combines a PEEK body with ATEC’s patented TiTec (titanium) coating technology to take advantage of the characteristics of both materials. The PEEK material allows surgeons to assess fusion through the implant while the titanium-coating provides initial stability due to the roughened ...
Manufactured by:ATEC Spine, Inc based inCarlsbad, CALIFORNIA (USA)
ATEC’s IdentiTi Posterior Straight Porous Ti Interbody Implants are designed to provide the biological, biomechanical, and imaging characteristics that surgeons seek in a fusion construct. The subtractive process used to manufacture each IdentiTi Implant results in more predictable mechanical performance and enhanced imaging ...
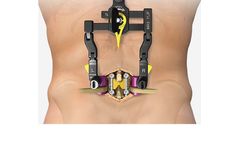
Manufactured by:NuVasive, Inc. based inSan Diego, CALIFORNIA (USA)
The system features an access design that can be adjusted to provide decompression, and integrates with our comprehensive fixation, biologics, and interbody implant portfolio, featuring a wide range of expandable and next-generation static cages. ...
Manufactured by:ChoiceSpine LLC based inKnoxville, TENNESSEE (USA)
This 510 (k) cleared device has a low-profile designed to be minimally invasive and yields less overhang than traditional plates, and can be inserted simultaneously with cervical interbody implants such as Tiger Shark™ C and Stealth™, thus eliminating surgical steps and potentially reducing operating room ...
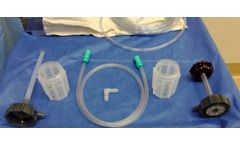
Manufactured by:Hensler Surgical Products based inWilmington, NORTH CAROLINA (USA)
The kit includes ergonomic containers and custom tubing, facilitating easy integration into surgical practices. Its design ensures that graft placement into interbody implants or packing around hardware is seamless. The HBP enhances the use of the patient's own osteoinductive and osteoconductive cells, integrating them into the fusion mass, and offering potential ...
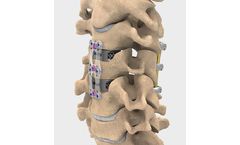
Manufactured by:NuVasive, Inc. based inSan Diego, CALIFORNIA (USA)
The thinnest plate system on the market*. The NuVasive anterior cervical plate (ACP) system is designed to achieve your ACDF surgical goals and help reduce dysphagia, malalignment and adjacent level ossification. ACP is procedurally integrated with versatile instrumentation, a complete interbody portfolio featuring advanced porous materials, traditional PEEK and allograft, biologics, the Maxcess ...