Refine by
Applications
- Prefilled Syringes and Combination Products for Nitrogen Dioxide Gas Sterilization
- Solutions for Pharmaceutical
- Microbial Reduction Solutions for Cosmetic and Packaging
- Microbiome Therapeutics for Patients and Physicians
- Flagship products for therapeutic industry
- Holter Blood Pressure and ECG Monitors for OEM
- Acoustic Bioprinter for Bridging from Bench to Bedside
- Acoustic Bioprinter for In Vitro Vascularization Controlled by Local Cell Density Enhancemen
- Receptor Antagonist Antiemetic Drug for Gastroenteritis & Gastritis
- Exosomes - Extracellular Vesicles for Validation and Quality Control
- Exosomes - Extracellular Vesicles for Cancer Treatment
- Exosomes - Extracellular Vesicles for Immunomodulating Response
- Exosomes - Extracellular Vesicles for Molecular Therapy
- Exosomes - Extracellular Vesicles for Brain Drug Delivery
- Exosomes - Extracellular Vesicles for Ocular Drug Delivery
Drug Production Equipment & Supplies In Libya
151 equipment items found
Manufactured by:Proveris Scientific Corporation based inHudson, MASSACHUSETTS (USA)
SprayVIEW Measurement Systems by Proveris are industry-standard tools designed to evaluate the spray patterns and plume geometry of orally inhaled and nasal drug products (OINDPs). These systems are crucial for both development and quality control phases, ensuring compliance with FDA guidelines and providing bioequivalence assessments. The system's adaptability ...
Manufactured by:Noxilizer, Inc based inHanover, MARYLAND (USA)
Compared to other sterilization methods, NO2 does not leave harmful residuals behind that could be cytotoxic. Pre-filled syringes and drug-delivery products administered in the sterile field and drugs with specific indications benefit most from NO2 sterilization. In two recent studies comparing NO2, EO and H2O2, results indicate the NO2 ...
Manufactured by:Noxilizer, Inc based inHanover, MARYLAND (USA)
Compared to other sterilization methods, NO2 does not leave harmful residuals behind that could be cytotoxic. Pre-filled syringes and drug-delivery products administered in the sterile field and drugs with specific indications benefit most from NO2 sterilization. In two recent studies comparing NO2, EO and H2O2, results indicate the NO2 ...
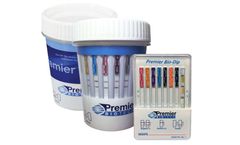
Manufactured by:Premier Biotech, Inc. based inMinneapolis, MINNESOTA (USA)
Premier Biotech’s CLIA-Waived (Clinical Laboratory Improvement Amendments) multi-drug integrated cup and dip products offer the ability to test for 5-14 drugs. CLIA-Waived test products are cleared by the FDA for over-the-counter (OTC) use, suited for workplace and clinical drug testing programs and ...
Manufactured by:BiosPacific, Inc. based inEmeryville, CALIFORNIA (USA)
Catalog number: D57330. product description: Amikacin Polyclonal Antibody. lot number: [lot specific]. immunogen: Amikacin. source: Sheep. concentration: [lot specific] mg/mL (based on A1%. 280nm = 14.0). storage: -20°C. hazard/biohazard: There is no known biohazard associated with this ...
Manufactured by:Cerbios-Pharma SA based inBarbengo – Lugano, SWITZERLAND
SF68® is considered one of the best-characterized probiotic active ingredients for the prevention and treatment of intestinal disorders, having a long history of safe use and proven efficacy. The original strain (Enterococcus faecium NCIMB 10415) was isolated in a healthy newborn baby in Sweden in the sixties, and selected for further use thanks to its favorable probiotic characteristics. The ...
Manufactured by:SureScreen Diagnostics Ltd. based inDerby, UNITED KINGDOM
Since the first use of fingerprint identification in 1858, we've known that the combination of whorls, arches and loops that make up the pattern of our fingerprint is unique to each ...
Manufactured by:SureScreen Diagnostics Ltd. based inDerby, UNITED KINGDOM
The biological content of oral fluid is directly relational to blood plasma. Therefore, if we detect drug metabolite in oral fluid, those metabolites are still present in the blood stream, indicating that the substance has recently been active in the body; so oral fluid is the choice matrix for ‘for-cause’ testing, but can also be used for random and pre-employment situations where ...
Manufactured by:SureScreen Diagnostics Ltd. based inDerby, UNITED KINGDOM
Drug metabolites begin appearing in urine a couple of hours after a substance is taken. These metabolites can be detectable in a urine sample for several days and up to 30 days after use. Whether using a dip or cup screening test, urine presents the widest range of parameters for drug screening, and is often the first matrix to feature new parameters. With best practice procedures in place, urine ...
Manufactured by:Haemopharm Healthcare S.r.l. based inMilano, ITALY
CSTD CSTD Refill® KIT, is the latest generation of systems for the reconstitution and administration of drugs, which consists of the combination of a dedicated bag and one or more Refill connectors for each vial to be reconstituted. In the common clinical practice of drug reconstitution, healthcare professionals perform a multi-step procedure that exposes ...
Manufactured by:Haemopharm Healthcare S.r.l. based inMilano, ITALY
The toxic effects of antineoplastic drugs used for cancer treatment, beyond the patient safety concerns, bring the occupational risks to health care workers handling hazardous drugs in the course of their duties. Multiple international studies(1) have documented, that healthcare workers who handle cytotoxic drugs have an increased risk of ...
Manufactured by:Allergan Aesthetics based inIrvine, CALIFORNIA (USA)
Most of the SkinMedica® products described on this website are intended to meet the FDA’s definition of a cosmetic product, an article applied to the human body to cleanse, beautify, promote attractiveness, and alter appearances. These SkinMedica® products are not intended to be drug products that ...
Manufactured by:Precision NanoSystems based inVancouver, BRITISH COLUMBIA (CANADA)
Combined with PNI’s expertise, we accelerate the clinical and commercial development of your unique nanomedicine drug ...
Manufactured by:Cambrex Corporation based inEast Rutherford, NEW JERSEY (USA)
Our drug product manufacturing and packaging capabilities cover a wide range of specialist dosage forms. We specialize in solid dose, liquids, gels, creams, ointments and sterile ointments and gels. For each of these dosage forms, we can provide pharmaceutical development through registration, as well as a full spectrum of commercial manufacturing services. ...
Manufactured by:Copley Scientific Ltd based inColwick, UNITED KINGDOM
Copley offers a range of testing equipment for additional qualification and validation of orally inhaled and nasal drug products (OINDPs), including assessments of generic drug formulations, inhaled dissolution testing and improving IVIVCs. View Our Inhaler Testing Brochure at: ...
Manufactured by:Adiso Therapeutics based inConcord, MASSACHUSETTS (USA)
ADS024 is an oral single strain live biotherapeutic product (SS-LBP). A naturally occurring live biotherapeutic product, ADS024 possesses a dual mechanism of action. It is bactericidal toward Clostridioides difficile and degrades Toxins A and B produced by C. difficile while maintaining other components of the microbiome. ADS024 is a single strain ...
Manufactured by:Cambrex Corporation based inEast Rutherford, NEW JERSEY (USA)
Our drug product manufacturing and packaging capabilities cover a wide range of specialist dosage forms. We specialize in solid dose, liquids, gels, creams, ointments and sterile ointments and gels. For each of these dosage forms, we can provide pharmaceutical development through registration, as well as a full spectrum of commercial manufacturing services. ...
Manufactured by:Multisorb based inBuffalo, NEW YORK (USA)
StabilOx oxygen absorbing canisters eliminate oxygen to help you achieve better product stability. This simple but effective solution a resides within packaging instead of the drug product itself, saving you from expensive drug product formulation changes. The science behind this carefully developed solution ...
Manufactured by:Newtech Medical Devices based inFaridabad, INDIA
The EvroPlus Evrolimus eluting stent system is a combination product comprised of two regulated components : a device (a coronary stent system) and a drug product (a formulation of Everolimus contained in a polymer coating) pre mounted a balloon catheter between two GOLD radio opaque marker ...
Manufactured by:Adiso Therapeutics based inConcord, MASSACHUSETTS (USA)
ADS024 is an oral single strain live biotherapeutic product (SS-LBP). A naturally occurring live biotherapeutic product, ADS024 modulates inflammation as a single strain multimodal LBP and has been shown to reduce neutrophil trafficking through the colonic epithelium. ADS024 is manufactured from a pure, clonal bacterial cell bank, yielding a standardized ...