Refine by
Locations
- USA
- Alabama
- Alaska
- Arizona
- Arkansas
- California
- Colorado
- Connecticut
- Delaware
- District Of Columbia
- Florida
- Georgia (USA)
- Hawaii
- Idaho
- Illinois
- Indiana
- Iowa
- Kansas
- Kentucky
- Louisiana
- Maine
- Maryland
- Massachusetts
- Michigan
- Minnesota
- Mississippi
- Missouri
- Montana
- Nebraska
- Nevada
- New Hampshire
- New Jersey
- New Mexico
- New York
- North Carolina
- North Dakota
- Ohio
- Oklahoma
- Oregon
- Pennsylvania
- Rhode Island
- South Carolina
- South Dakota
- Tennessee
- Texas
- Utah
- Vermont
- Virginia
- Washington
- West Virginia
- Wisconsin
- Wyoming
Disease Risk Equipment & Supplies In USA
42 equipment items found
Manufactured by:Arkray USA, Inc. based inMinneapolis, MINNESOTA (USA)
Highly Accurate1 Blood Glucose Testing in a Feature-focused System. Tight glycemic control can reduce the risk of diabetes-related complications by at least 42%*.2. * 76% reduced risk of eye disease; 50% reduced risk of kidney disease; 60% reduced risk of nerve disease; 57% ...
Manufactured by:Randox Laboratories Ltd. based inCrumlin, UNITED KINGDOM
Randox is a leading provider of diagnostic reagents for the assessment of cardiovascular disease risk. Our extensive menu of cardiac biomarkers within the cardiology reagents panel include: routine lipid tests such as Homocysteine, hsCRP, Apo A-I, Apo A-II, Apo B and Lp(a), as well as, unique assays for cardiac risk assessment ...
Manufactured by:DTR Medical Ltd, part of the Innovia Medical Group based inSwansea, UNITED KINGDOM
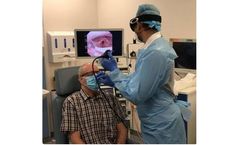
Protects from airborne risks and diseases. A one-way valve that can be retrofitted to any fluid resistant Surgical Mask. Designed with the aim to keep Clinicians safe and so better able to deliver high quality care to their patients. Facilitates the movement of a Nasendoscope up to 4mm in diameter. ...
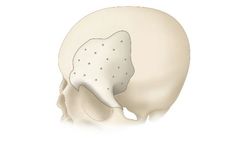
Manufactured by:Bioplate based inPlacentia, CALIFORNIA (USA)
Indicated for replacing bony voids in cranial and craniofacial skeletons, excluding maxillofacial regions, these implants avoid complications such as infection and poor blood supply, enhancing the reliability in surgical outcomes. It's crucial to avoid use in patients with systemic infections or significant degenerative diseases. Potential risks include material ...
Manufactured by:ELITechGroup based inPuteaux, FRANCE
Aspergillosis is the most common cause of fungal infection among stem cell and solid organ transplant recipients, resulting in high mortality if untreated. Patients with weakened immune system and lung diseases are at higher risk of developing severe outcomes. Early detection and quantification of Aspergillus with molecular methods is crucial to isolate infected ...
Manufactured by:Endodiag based inParis, FRANCE
Endodiag together with several expert gynecologists has developed EndoGram, a 10-biomarker test based on the analysis of biopsies taken during surgery. Thanks to a proprietary algorithm combining other patient clinical factors, EndoGram provides for each patient a risk of disease recurrence score and a biological profile aimed to help the physician discard ...
Manufactured by:Setpoint Medical Corporation based inValencia, CALIFORNIA (USA)
SetPoint Medical created a therapy to offer patients and providers a better alternative for the treatment of chronic inflammatory diseases with potentially less risk and lower cost than drug therapy. SetPoint Medical’s bioelectronic system is free of routine injections or pills and may eliminate the risks and substantial cost associated ...
by:Natera, Inc. based inAustin, TEXAS (USA)
Renasight is a test to determine if there is a genetic cause for an individual’s kidney disease or if there is an increased hereditary risk due to family history. The test uses a blood or saliva sample to test 385 genes associated with chronic kidney disease (CKD). Results are available in approximately 3 ...
Manufactured by:Medosis based inSofia, BULGARIA
Composed of a synthetic poly(lactic acid) (PLA) polymer known for its biocompatibility and bioresorbability, it features a fully resorbable profile, breaking down within 15-20 weeks post-implantation. This material mitigates any risk of disease transmission by avoiding human or animal tissues. The external membrane surface, made of non-porous PLA, prevents ...
Manufactured by:Cysview, Trademark of Photocure based inPrinceton, NEW JERSEY (USA)
Historically, bladder cancer patients are at high risk for disease recurrence and progression,1 which could be due in part to missed tumors and incomplete surgeries because not all cancerous tissue is easy to see under white light. Moving forward, with the availability of Blue Light Cystoscopy with Cysview, that trend may ...
Manufactured by:Biçakcilar based inÜsküdar, TURKEY
We are proud to introduce our new "Suction Catheters With Protective Sleeve”, designed to prevent aerosol spread of microorganisms into the nearby environment, and reducing risk of disease spread among our precious frontline healthcare professionals, doctors and nurses, engaged in a relentless war against COVID-19. Suction catheters with protective sleeve ...
by:LightDeck Diagnostics based inBoulder, COLORADO (USA)
We are developing point-of-care, multiplex tests for host-response biomarkers in ...
Manufactured by:Perrery Farmaceutici S.r.l. based inPescantina (VR), ITALY
Helicoter is a new approach in treatment of patients with H. pylori infection. Helicoter contains a free-flowing powder obtained through fermentation and spray-drying of Lactobacillus reuteri DSM 17648, which has a specific anti-helicobacter effect. It specifically binds and aggregates the stomach bacterium Helicobacter pylori. Thanks to this unique mode of action, Helicoter helps to reduce the ...
Manufactured by:Acera Surgical Inc. based inLouis, MISSOURI (USA)
For surgeons who need a dural repair solution, Cerafix is a non-biologic, implantable, bioresorbable, fibrous matrix engineered to offer reliable dural closure, reduce postoperative cerebrospinal fluid leakage (CSF), and support dural ...
Manufactured by:Pathnostics based inIrvine, CALIFORNIA (USA)
This test identifies genetic abnormalities in patients with Barrett’s esophagus and provides an indication of progression requiring additional procedures and targeted management. Utilizing cytology, immunohistochemistry (IHC), and fluorescence in situ hybridization (FISH) testing on brushings from patients with Barrett’s esophagus, patient chromosomal alterations can be distinguished ...
Manufactured by:Ariana Pharma based inParis, FRANCE
Ariana® has consolidated multiple, publicly available administrative healthcare admissions databases into a locally searchable observational health data resource of patient hospitalization, outpatient and other episodes, comprising in excess of 6B records. ...
Manufactured by:Vitrosens Biotechnology A.S. based inÜmraniye, TURKEY
It is created for in vitro quantitative detection of the concentration of C-reactive (CRP) and hypersensitive CRP (hsCRP) proteins in human serum, plasma, and whole blood samples. Automated and objective results in 3 minutes by immunofluorescent assay. ...
Manufactured by:Xinle Huabao Medical Products Co., Ltd. based inXinle, CHINA
Disposable medical protective coverall is applicable to GB19082 and EN14126+Type 5b+Type 6B. It can provide barrier and protection for clinical medical personnel to contact with potentially infectious patients' blood, body fluids, secretions, particles in the air, etc. the product is made of microporous PE film and high Anti-static non-woven composite materials, with soft hand feeling, ...
Manufactured by:Cabaletta Bio, Inc. based inPhiladelphia, PENNSYLVANIA (USA)
DSG3/1-CAART is being developed to treat mucocutaneous pemphigus vulgaris (mcPV), a B cell-mediated autoimmune disease that causes blistering of the skin and mucous membranes. mcPV is caused by autoantibodies that attack the cell adhesion proteins DSG3 and DSG1. DSG3/1-CAART is designed to selectively target and kill the B cells that express the pathogenic autoantibodies while preserving the ...
Manufactured by:The Electrospinning Company Ltd. based inDidcot, UNITED KINGDOM
The Electrospinning Company is developing the Symatix® membrane which is an innovative synthetic amniotic membrane substitute. The membrane is created using electrospinning technology and is designed to overcome the limitation of the Human Amniotic Membrane ...