Show results for
Refine by
Antigen Protein Equipment & Supplies In Mali
60 equipment items found
Manufactured by:ImevaX GmbH based inMunich, GERMANY
(flagellar filament capping protein or flagellar hook-associated protein) is an essential element in the assembly of H. pylori´s flagella. It is an efficient, highly sensitive and specific marker, present in all H. pylori strains worldwide. ImevaX holds the patent on the detection of FliD-specific ...
Manufactured by:Oy Medix Biochemica Ab based inEspoo, FINLAND
Recombinant human pepsinogen I ...
Manufactured by:Beijing Meizheng Bio-Tech Co., Ltd based inYanqing District, CHINA
Glutathione S-transferase (GST) Gene Fustion System is a versatile system for the expression, purification, and detection of GST-tagged proteins produced in E. coli. Purification of GST-tagged proteins can be performed under very mild conditions, which preserves the function and antigenicity of the target ...
Manufactured by:Response Biomedical Corp. based inVancouver, BRITISH COLUMBIA (CANADA)
Response Biomedical provides a test that aids in the rapid diagnosis of RSV infections in symptomatic patients 21 years of age and younger. It detects RSV F-protein antigens in nasal wash, NP swab or NP aspirate specimens. It provides an objective and qualitative results in ~15 minutes. ...
Manufactured by:Novodiax, Inc. based inHayward, CALIFORNIA (USA)
Cluster of Differentiation 45 (CD45) pHRP Anti-Human Staining Kit immunohistochemistry (IHC) reagent is for use on frozen and formalin-fixed-paraffin-embedded tissues. The CD45 antigen is a protein which was originally called Leukocyte Common Antigen and recognizes an antigen found on lymphoid cells. Anti-CD45 antibody is a ...
Manufactured by:Wuhan HealthCare Biotechnology Co., Ltd. based inWuhan, CHINA
For qualitative detection of the nucleocapsid protein antigen of SARS-CoV-2 in nasopharyngeal and nasal swab, and saliva specimens from individuals and is aid in rapid diagnosis of patients with suspected SARS-CoV-2 ...
Manufactured by:CTK Biotech, Inc. based inPoway, CALIFORNIA (USA)
Carcinoembryonic antigen is a small protein or antigen found in tissues of developing fetus, then becomes very low or disappears after birth. The normal range of CEA levels in healthy adults is 0 – 2.5 ng/ml. CEA levels increase in the blood of some cancer patients as CEA is found on the surface of many types of cancer cells. ...
Manufactured by:Mountain Horse Solution based inUnion City, TENNESSEE (USA)
The Flowflex COVID-19 Antigen Home Test is a rapid test for the detection of SARS-CoV-2. The lateral flow chromatographic immunoassay is intended for the qualitative detection of the nucleocapsid protein antigen from SARS-CoV-2 in anterior nasal swab specimens directly from individuals within 7 days of symptom onset or without symptoms or other ...
Manufactured by:Gold Standard Diagnostics based inDavis, CALIFORNIA (USA)
The Antigen Rapid Test assay from the Gold Standard Diagnostics Group is a rapid chromatographic immunoassay for the qualitative detection of SARS-CoV-2 Nucleocapsid (N) Protein antigens present in respiratory ...
Manufactured by:ACROBiosystems based inNewark, DELAWARE (USA)
Name:Receptor protein-tyrosine kinase erbB-2. Target Synonym:EC 2.7.10.1,HER-2/Neu,Herstatin,HER-2,TKR1,Receptor, ErbB-2,V-Erb-B2 Erythroblastic Leukemia Viral Oncogene Homolog 2, Neuro/Glioblastoma Derived Oncogene Homolog,V-Erb-B2 Avian Erythroblastic Leukemia Viral Oncoprotein 2,Neuroblastoma/Glioblastoma Derived Oncogene Homolog,C-Erb B2/Neu Protein,CD340 ...
Manufactured by:Access Bio based inSomerset, NEW JERSEY (USA)
Self Test for SARS-CoV-2 Antigen Detection. The CareStart™ COVID-19 Antigen Home Test is a lateral flow immunoassay intended for the qualitative detection of nucleocapsid protein antigens from SARS-CoV-2. Fast and easy to self-test at anywhere. Easy to interpret the results using mobile application. Qualitatively detect the ...
Manufactured by:Genabio Diagnostics Inc. based inBedford, MASSACHUSETTS (USA)
Intended use: iHealth® COVID-19 Antigen Rapid Test is a lateral flow assay intended for the qualitative detection of nucleocapsid protein antigen from SARS-CoV-2. This test is authorized for non-prescription home use with self-collected anterior nasal (nares) swab samples from individuals aged 15 years or older with symptoms of COVID-19 ...
Manufactured by:VBI Vaccines Inc. based inCambridge, MASSACHUSETTS (USA)
Because of their structural similarity to viruses presented in nature, including their particulate nature and repetitive structure, virus-like particles (VLPs) are ideal for stimulating potent immune responses. VLPs can be customized to present any protein antigen, including multiple antibody and T cell targets, making them ideal technologies for the development ...
Manufactured by:Novodiax, Inc. based inHayward, CALIFORNIA (USA)
Cluster of Differentiation 45 (CD45) pHRP Anti-Human Staining Kit immunohistochemistry (IHC) reagent is for use on frozen and formalin-fixed-paraffin-embedded tissues. The CD45 antigen is a protein which was originally called Leukocyte Common Antigen and recognizes an antigen found on lymphoid cells. Anti-CD45 antibody is a ...
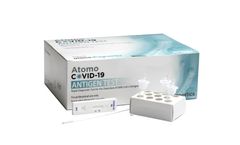
Manufactured by:Atomo Diagnostics Limited based inLeichhardt, AUSTRALIA
A fast, accurate point of care COVID-19 antigen test for the qualitative detection of nucleocapsid protein antigens specific to the SARS-CoV-2 virus. Utilising an anterior nasal swab specimen obtained from individuals suspected of having COVID-19, the Atomo COVID-19 Antigen Test is easy to use, giving a result in as little as 10 ...
Manufactured by:Lumos Diagnostics based inSarasota, FLORIDA (USA)
CoviDx™ SARS-CoV-2 Rapid Antigen Test is a lateral flow assay intended for the qualitative detection of the nucleocapsid protein antigen from SARS-CoV-2 in nasal swabs and nasopharyngeal swabs from patients suspected of a Coronavirus-19 (COVID-19) infection by their healthcare provider within the first 5 days of ...
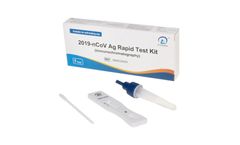
Manufactured by:ulti med Products (Deutschland) GmbH based inAhrensburg, GERMANY
This kit is used for the in vitro qualitative detection of 2019-nCoV antigen. It is an immunochromatography sandwich assay, and intended to detect 2019-nCoV N-protein antigen in human nasal (NS) swab specimens. This kit can be used for individuals with or without symptoms or other epidemiological reasons to suspect COVID-19 ...
Manufactured by:Genabio Diagnostics Inc. based inBedford, MASSACHUSETTS (USA)
Intended Use: INDICAID™ COVID-19 Rapid Antigen Test is a lateral flow immunoassay intended for the qualitative detection of nucleocapsid protein antigen from SARSCoV-2 in direct anterior nasal swab specimens from individuals who are suspected of COVID-19 by their healthcare provider within the first five (5) days of symptom onset. ...
Manufactured by:10x Genomics based inPleasanton, PENNSYLVANIA (USA)
Chromium Single Cell Immune Profiling provides a multiomic solution to your immunology questions. Analyze full-length, paired B-cell or T-cell receptors, surface protein expression, antigen specificity, and gene expression, all from a single cell. ...
Manufactured by:Aesku.Group GmbH based inWendelsheim, GERMANY
The AESKU.RAPID SARS-CoV-2 Rapid Test is based on immunochromatographic polymer technology combined with the sandwich principle for the qualitative detection of the nucleocapsid protein antigen in human nasal swab specimens. The specimen is placed in the test device's specimen well with colored polymer-labeled monoclonal SARS-CoV-2 antibody 1 and mixed along the ...