Show results for
Refine by
Locations
- USA
- Alabama
- Alaska
- Arizona
- Arkansas
- California
- Colorado
- Connecticut
- Delaware
- District Of Columbia
- Florida
- Georgia (USA)
- Hawaii
- Idaho
- Illinois
- Indiana
- Iowa
- Kansas
- Kentucky
- Louisiana
- Maine
- Maryland
- Massachusetts
- Michigan
- Minnesota
- Mississippi
- Missouri
- Montana
- Nebraska
- Nevada
- New Hampshire
- New Jersey
- New Mexico
- New York
- North Carolina
- North Dakota
- Ohio
- Oklahoma
- Oregon
- Pennsylvania
- Rhode Island
- South Carolina
- South Dakota
- Tennessee
- Texas
- Utah
- Vermont
- Virginia
- Washington
- West Virginia
- Wisconsin
- Wyoming
Bone Void Equipment Supplied In USA
30 equipment items found
Manufactured by:EnColl Corporation based inFremont, CALIFORNIA (USA)
High purity Type-I collagen derived from bone is essential for tissue regeneration and remodeling in any osseous defect. Osseograft / DMBM (xenograft) is one such de-mineralized bone derived Type-I collagen for bone void filling ...
Manufactured by:EnColl Corporation based inFremont, CALIFORNIA (USA)
Over 100 years of research, experimentation and use has demonstrated that calcium sulphate enhances bone regeneration and is totally biocompatible and and resorbable. At the same token, high purity type-I collagen DMBM (xenograft) also is essential for tissue regeneration and remodeling. Osseomold consists of both materials admixed in proper proportions to give the best biological and mechanical ...
Manufactured by:Parametrics Medical based inLeander, TEXAS (USA)
Synthetic bone void fillers are safe, biocompatible, and easy to use. These resorbable biomaterials are based on nano-crystalline hydroxyapatite (HA) and purified collagen technologies. These products are engineered to optimize the resorption rate. Our granules are composed of 60% HA and 40% β-Tricalcium Phosphate (TCP). The formulation provides optimal ...
Manufactured by:OrthoPediatrics Corp. based inWarsaw, INDIANA (USA)
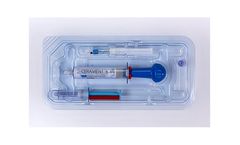
CERAMENT BONE VOID FILLER is an injectable, moldable and drillable synthetic bone void filler consisting of 40% hydroxyapatite, 60% calcium sulfate and the radio-contrast agent iohexol. The unique ratio of hydroxyapatite and calcium sulfate is designed to enable CERAMENT to resorb at the same rate that bone ...
Manufactured by:Parametrics Medical based inLeander, TEXAS (USA)
OsteoSource™ allograft bone particulates have osteoconductive properties and are well suited to fill bone structure defects. Osteoconductive allografts act as a scaffold for the growth of natural bone. Over time, the natural bone replaces the donor’s bone. OsteoSource™ eliminates the need for autograft bone, associated with donor site morbidity, longer surgery time, and ...
Manufactured by:SurgiLogix based inSan Antonio, TEXAS (USA)
SXDBM is an advanced healing technology that is revolutionizing the way doctors approach bone defects. This unique human allograft tissue is derived from human bone which has undergone a specialized demineralization process that maintains the bone’s natural growth factors. When implanted into a bone void or ...
Manufactured by:Zetagen Therapeutics Inc. based inSyracuse, NEW YORK (USA)
ZetaBase is an osteo-conductive and osteo-promotive therapy and is intended for the use as a bone void filler for bony voids or gaps that are not intrinsic to the stability of the bony structure. ZetaBase Bone Graft can be used as a bone graft ...
by:In2Bones Global based inMemphis, TENNESSEE (USA)
The i2b Resorbable Bead Kit is designed to create easy-to-use bio-resorbable beads for filling osseous defects. i2b® Resorbable Bead Kits allow surgeons to cast 100% pure synthetic resorbable bone void filler Beads in two sizes: 3.5 and 4.5mm. The beads provide a safe alternative to human or animal cadaver bone that completely eliminates the ...
Manufactured by:VIVEX Biologics, Inc. based inMiami, FLORIDA (USA)
VIA DBM Plus™ is a demineralized bone matrix putty containing cortical bone chips and indicated for use in the extremities, posterolateral spine, and pelvis as a bone void filler or for treatment of osseous ...
Manufactured by:Surgentec LLC based inBoca Raton, FLORIDA (USA)
Surgentec brings a new advancement to the bone graft delivery arena with the Graftgun. Our Graft Delivery System (GDS®) is designed to allow for universal, quick, and accurate bone graft delivery to a surgical site without the problems of a traditional funnel.Its patented, controlled delivery method is designed to safely dispense bone graft ...
Manufactured by:Spine Wave, Inc. based inShelton, CONNECTICUT (USA)
Tempest® Allograft Bone Matrix is a sterile, ready-to-use allograft bone matrix composed of cancellous and partially demineralized cortical bone for use in filling a variety of bone voids. Its proprietary processing yields a bone allograft product with unique handling characteristics that is ...
by:Molecular Matrix, Inc. based inWest Sacramento, CALIFORNIA (USA)
Osteo-P™ Bone Graft Substitute (BGS) is a non-mineralized, synthetic bone void filler consisting of a hyper-crosslinked carbohydrate polymer. It is highly porous, biocompatible, biodegradable, and osteoconductive. Osteo-P™ BGS is intended for the filling of bone defects created surgically or as a result of traumatic ...
Manufactured by:Xenco Medical based inSan Diego, CALIFORNIA (USA)
The Sorrento™ Bone Graft Substitute is a robust, resorbable bone void filler to fill voids or gaps of the skeletal system in the extremities and pelvis not intrinsic to the stability of the bony structure. Pre-clinical animal studies have revealed its tremendous performance through early osteoblast activity and osteocyte ...
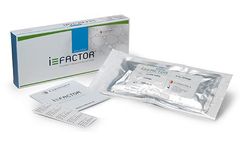
Manufactured by:Cerapedics, Inc. based inWestminster, COLORADO (USA)
i-FACTOR Bone Graft products are intended to replace or augment the use of autologous bone commonly utilized in spine, trauma, and orthopedic procedures such as: Spinal fusion incorporating interbody fusion devices; Treatment of non-union or traumatic fresh fractures; Joint reconstruction requiring a bone void filler. i-FACTOR ...
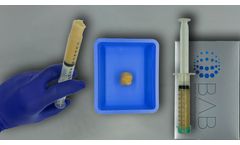
Manufactured by:Berkeley Advanced Biomaterials (BAB) based inBerkeley, CALIFORNIA (USA)
H-GENIN Putty is intended for use as a demineralized bone matrix for voids or gaps that are not intrinsic to the stability of the bony structure. Putty is indicated for the treatment of surgically created bone defects or bone defects created from traumatic injury. H-GENIN Putty provides a moldable consistency and can be mixed ...
Manufactured by:Berkeley Advanced Biomaterials (BAB) based inBerkeley, CALIFORNIA (USA)
H-GENIN Crush-Mix is intended for use as a demineralized bone matrix for voids or gaps that are not intrinsic to the stability of the bony structure. Putty is indicated for the treatment of surgically created bone defects or bone defects created from traumatic injury. H-GENIN Crush-Mix provides a moldable consistency and can be ...
Manufactured by:Safeguard Medical based inHuntersville, NORTH CAROLINA (USA)
The NIO Infant™ provides safe, rapid intraosseous access in the proximal tibia as an alternative to IV access in emergency medical situations. The NIO Infant is a manual IO device with an innovative Stepped Needle® which provides graduated penetration into the bone marrow cavity to help prevent over-penetration. Additionally, the NIO Infant includes a Fixation dressing, which secures ...
Manufactured by:Kinamed Incorporated based inCamarillo, CALIFORNIA (USA)
A simple set of instruments allows for precise creation of two compound wedges of bone. Transposing these wedges medializes the tubercle. ...
Manufactured by:Cervos Medical based inMarshfield, MASSACHUSETTS (USA)
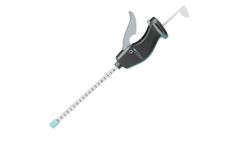
The CER-SUB-1560 is a bone access device that has a 15 gauge cannula with an open tip and is designed to be powered by a drill. The device can serve as an access port to transplant tissue or medications. For example, marrow aspirate obtained using CER-EXT can be used with the CER-SUB-1560 to transplant marrow aspirate. The mechanism your body uses to repair and maintain tissue is Vasculogenesis, ...
Manufactured by:Osteogenics Biomedical based inLubbock, TEXAS (USA)
Zcore™ is an osteoconductive, porous, anorganic bone mineral with a carbonate apatite structure derived from porcine cancellous bone. Proprietary processing steps preserve both interconnecting macroscopic and microscopic porous structures of Zcore™ that helps support the formation and in-growth of new bone. ...