Refine by
Clinical Development Equipment & Supplies In Belize
206 equipment items found
Manufactured by:HepaRegeniX GmbH based inTübingen, GERMANY
HepaRegeniX has started to progress the clinical development of its proprietary candidates e.g. ...
by:Vivozon Inc. based inYongin-si, SOUTH KOREA
A randomized, double-blind, placebo-controlled, single and repeated dose, escalation clinical trial to evaluate the safety, tolerability, and pharmacokinetics of VVZ-149 injection in healthy adult ...
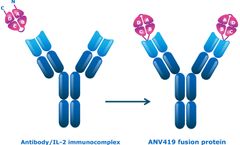
Manufactured by:Anaveon AG based inBasel, SWITZERLAND
The culmination of our research is ANV419, which is in clinical development. It is a rationally engineered fusion protein that is a stable, antibody-like molecule. ...
Manufactured by:Aristotech Industries GmbH based inLuckenwalde, GERMANY
The Design of POROCUP is based on the principles of the well-known and long term clinically proven TITANIUM CUP developed in Switzerland in the 90´s. Applying a Titanium Forging Technology, a macro porosity surface is created, which favors the bone osteointegration without the necessity of additional ...
Manufactured by:Apyx Medical Corporation based inClearwater, FLORIDA (USA)
Renuvion cosmetic technology is the result of extensive scientific research and clinical development. Because of its proprietary balance of helium and RF energy, Renuvion has been described as ideal for medical procedures where precision and control are key to an effective ...
by:Vivozon Inc. based inYongin-si, SOUTH KOREA
Single-dose, escalation clinical trial to evaluate the safety and pharmacokinetics of VVZ-149 injection in healthy elderly ...
Manufactured by:CytomX Therapeutics, Inc. based inSouth San Francisco, CALIFORNIA (USA)
CytomX and Amgen are developing CX-904, a T-cell engaging bispecific Probody candidate against Epidermal Growth Factor Receptor (EGFR) and CD3. The drug candidate is advancing towards IND-enabling studies. CytomX is responsible for the IND filing, targeted for late 2021, and for early clinical development with Amgen leading later stage development and ...
Manufactured by:Precision NanoSystems based inVancouver, BRITISH COLUMBIA (CANADA)
Built on our revolutionary NxGen™ technology found in our NanoAssemblr® family of instruments, NanoAssemblr® GMP System enables you to go from concept to clinic with speed and confidence. Combined with PNI’s expertise, we accelerate the clinical and commercial development of your unique nanomedicine drug ...
Manufactured by:Aristotech Industries GmbH based inLuckenwalde, GERMANY
Femoral Stem Forgings based on the design concepts of clinically proven uncemented stems, developed in the ...
by:Vivozon Inc. based inYongin-si, SOUTH KOREA
A randomized, double-blind, parallel-group, placebo-controlled clinical trial to evaluate the analgesic efficacy and safety of VVZ-149 Injection in patients who have pain after laparoscopic and robotic-laparoscopic ...
by:Vivozon Inc. based inYongin-si, SOUTH KOREA
A multicenter, randomized, double-blind, parallel-group, placebo-controlled phase 3 clinical trial to evaluate the analgesic efficacy and safety of VVZ-149 injection in patients who have pain after laparoscopic ...
Manufactured by:Spectral Medical Inc. based inToronto, ONTARIO (CANADA)
Superior reagents are key to developing exceptional clinical diagnostic products and Spectral continues to provide high quality, high performance biochemicals for research and commercial products. Possessing a long onboard shelf life, Spectral’s single-chain Troponin I-C polypeptides, and other reagent products, have been recognized by the market as a premier reagent for large instrument ...
Manufactured by:Shenzhen SONTU Medical Imaging Equipment Co., Ltd based inShenzhen, CHINA
Adhering to the ergonomic design, high-end manufacturing technology, and strict testing process, Polaris Series U-arm DR with a professional image chain can meet the needs of multiple clinical ...
Manufactured by:Genenta Science based inMilano, ITALY
After apheresis collection, a functional copy of the therapeutic gene is inserted into the patient’s own HSPCs using a non-replicating lentiviral vector. This is an ex-vivo process called transduction. We believe that LVVs are the first choice for ex-vivo gene therapy in humans because they can efficiently transduce HSPCs with potentially large transgenes that will allow us to expand ...
Manufactured by:Creative Peptides based inNew York, NEW YORK (USA)
Abaloparatide is a parathyroid hormone-related protein (PTHrP) analog drug in clinical development for treating osteoporosis. Like the related drug teriparatide, and unlike bisphosphonates, it is an anabolic (i.e., bone growing) agent.https://www.creative-peptides.com/product/abaloparatide-item-10-101-172-33658.html ...
Manufactured by:Aurinia Pharmaceuticals Inc. based inVictoria, BRITISH COLUMBIA (CANADA)
AUR200 is a recombinant Fc fusion protein designed to specifically block B-cell Activating Factor, known as BAFF, and A Proliferation-Inducing Ligand, known as APRIL. BAFF and APRIL promote B cell survival and differentiation and have been shown to play a prominent role in the pathogenesis of certain autoimmune and nephrology conditions. AUR200 is currently undergoing pre-clinical development ...
Manufactured by:Interacoustics based inMiddelfart, DENMARK
A versatile device offering auditory evoked potentials (AEP), auditory steady state response (ASSR), vestibular evoked myogenic potentials (VEMP) and otoacoustic emissions (OAE). Designed to fit seamlessly into your workflow and offers complete reliability and perfect results. ...
by:STALICLA based inGeneve, SWITZERLAND
DDU validates ASD phenotype groups and drug candidates through observational studies and investigational trials. DDU combines expertise in operations, medicine, drug and clinical development, to bring STALICLA’s clinical-stage treatment options closer to ...
Manufactured by:BioArctic AB based inStockholm, SWEDEN
The drug candidate ABBV-0805 is a monoclonal antibody that selectively binds and eliminates oligomers and protofibrils of alpha-synuclein. The goal is to develop a disease modifying treatment that stops or slows down disease ...
Manufactured by:Illumina Inc. based inSan Diego, CALIFORNIA (USA)
The NextSeq 550Dx instrument is FDA regulated and CE-in vitrodiagnostic (IVD) marked, enabling clinical laboratories to develop and perform a wide range of applications, from NGS IVD assays using targeted panels, to clinical research applications that include methods from targeted panels to whole ...