Refine by
Clinical Program Equipment & Supplies In Spain
44 equipment items found
Manufactured by:OSE Immunotherapeutics based inNantes, FRANCE
A humanized monoclonal antibody, is an antagonist of the alpha chain of the interleukine-7 receptor, CD127, present on T effector cells, thus down regulating the immune activity. Interleukin-7 (IL-7) is a cytokine that controls the proliferation, apoptosis and activation of CD4 and CD8 effector T-cells in ...
Manufactured by:OSE Immunotherapeutics based inNantes, FRANCE
a monoclonal antibody fragment and CD28 antagonist, selectively blunts CD28 co-stimulation while sparing the CTLA-4 co-inhibitory signal. The net effect of CD28 antagonism is down regulating effector T-cells while promoting T-Reg activity. The CD28/CTLA-4 pathway controls both T-cell activation and regulatory T-cell down regulation. CD28 receptor is expressed on the surface of the majority of ...
Manufactured by:OSE Immunotherapeutics based inNantes, FRANCE
In recent years, immunotherapies have driven significant clinical progress and increased hopes for patients suffering from various types of cancer. The tumor microenvironment (TME), in particular the presence and immune-status of myeloid derived suppressor cells (MDSC), tumor-associated macrophages (TAM) as well as dendritic cells, has been closely linked to tumor activity and responsiveness to ...
Manufactured by:OSE Immunotherapeutics based inNantes, FRANCE
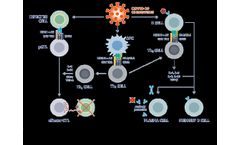
CoVepiT, a multi-target and multi-variant vaccine candidate against COVID-19. CoVepiT is a next-generation multi-target, multi-variant vaccine against SARS-CoV-2 in clinical Phase 1. The vaccine candidate was designed using optimized epitopes selected after screening more than 67,000 global SARS-CoV-2 genomes, as well as those of previous human-infective CoVs, SARS and MERS, to identify vaccine ...
Manufactured by:OSE Immunotherapeutics based inNantes, FRANCE
Tedopi®, OSE Immunotherapeutics’s most advanced product and therapeutic neo-epitope-based vaccine, is a proprietary combination of nine optimized neo-epitopes, selected and optimized from 5 tumoral antigens to activate specifically T cells, plus one epitope giving universal helper T cell response targeting T cell ...
Manufactured by:Elicio Therapeutics based inBoston, MASSACHUSETTS (USA)
Our lymph node-targeted mKRAS cancer vaccine; Our Amphiphile (AMP)-powered lead candidate, ELI-002, generates a robust immune response against mutant Kristen rat sarcoma (mKRAS), with the potential to offer patients a stronger fight against the most aggressive cancers. Preliminary data have shown that ELI-002 produces a potent mKRAS-specific T-cell response, including both CD4+ and CD8+ ...
Manufactured by:Aposcience AG based inVienna, AUSTRIA
Discussions are underway for the design of a clinical program in this indication together with a manufacturer of catheter devices suitable for direct myocardial injection of APO-1 ...
Manufactured by:AOBiome Therapeutic, LLC based inCambridge, MASSACHUSETTS (USA)
We have completed six Phase 2 clinical programs and multiple pre-clinical programs. Over 1,000 patients have been enrolled in our clinical trials since 2016. All of those patients except for 28 in our open label pediatric study have been enrolled in randomized double-blind placebo controlled ...
Manufactured by:Daiichi Sankyo Company Limited based inTokyo, JAPAN
Oral, once-daily, direct factor Xa (pronounced “Ten A”) inhibitor. More than 100,000 patients worldwide are expected to participate in the edoxaban clinical research program, which is comprised of more than 10 RCTs (randomized, controlled trials), non-interventional studies and registries, including completed, ongoing and future research. ...
Manufactured by:Elixir Medical Corporation based inMilpitas, CALIFORNIA (USA)
Sirolimus is the most widely known mTOR inhibitor with a well-established safety and effectiveness profile in various treatment applications: Drug-eluting stents, Transplant, Oncology. Novolimus and Sirolimus have been studied as part of Elixir Medical’s Clinical Programs. These clinical trials for DynamX™, DESyne®, ...
by:Verb Biotics based inBoston, MASSACHUSETTS (USA)
Yso1 is a clinical stage development program aiming at harnessing the potential of Christensenella minuta as a groundbreaking biotherapy to treating obesity and metabolic disorders. The goal of our disruptive approach is to return to obese patients christensenella, which have the unique ability to repair the unbalanced microbiome, thereby restoring impaired key ...
Manufactured by:Premier Biotech, Inc. based inMinneapolis, MINNESOTA (USA)
Premier Biotech’s CLIA-Waived (Clinical Laboratory Improvement Amendments) multi-drug integrated cup and dip products offer the ability to test for 5-14 drugs. CLIA-Waived test products are cleared by the FDA for over-the-counter (OTC) use, suited for workplace and clinical drug testing programs and approved for waiver under the CLIA ...
Manufactured by:Catalent, Inc based inSomerset, NEW JERSEY (USA)
Antibody-drug conjugates (ADCs) combine the targeted binding specificity and pharmacological advantages of monoclonal antibodies with the potency advantages of small molecule chemotherapy agents via conjugation with chemical linkers. With 10 approved products and a large and growing list of programs in clinical and pre-clinical development, ADCs ...
Manufactured by:RJL Systems, Inc based inClinton Township, MICHIGAN (USA)
Designed for professionals seeking a cost-effective and dependable solution, the Legacy provides accurate measurements of fat mass, lean mass, and body water—essential data for health assessments, wellness programs, and clinical ...
by:Align Technology, Inc. based inTempe, ARIZONA (USA)
The Invisalign system is the most advanced clear aligner system in the world. Backed by two decades of innovation, the Invisalign system has helped bring smiles to over 9.6 million people including over 2.4 million ...
Manufactured by:Omni-Biotic based inPort Chester, NEW YORK (USA)
This 3-month OMNi-BiOTiC® probiotic program is designed to reset the gut microbiome and support overall wellness. Based on over 30 years of gut microbiome research and 100+ clinical studies, this program is curated by the Founder of Omni-Biotic and CEO of the globally recognized microbiome science center Institut Allergosan, Anita ...
Manufactured by:Ito Co., Ltd. based inKawaguchi City, Saitama, JAPAN
Electrotherapy Device with 2 Independent Channels and Body Diagram ...
Manufactured by:Ito Co., Ltd. based inKawaguchi City, Saitama, JAPAN
Electrotherapy Device with 4 Independent Channels and Body Diagram ...
Manufactured by:SensitivE Audit based inStaten Island, NEW YORK (USA)
This module provides for obtaining the following: unbiased findings on the human body quantitative composition, linear control and timely correction of nutrition programs in clinical practice and sports medicine, an unbiased assessment of physical conditioning and nutrition, analysis of water-salt metabolism, analysis of the state of water-salt metabolism during ...
Manufactured by:Minaris Regenerative Medicine, LLC based inAllendale, NEW JERSEY (USA)
Having this team creates the potential to accelerate your progress to market and to both anticipate and overcome your challenges, while reducing the overall cost and risk of your clinical and commercial development programs. ...