Refine by
Applications
- SeeFactor CT3 for Consider Ear, Nose & Throat (ENT)
- SeeFactor CT3 for Consider Orthopedics (Hips, Knees, Ankles, Bone Density)
- SeeFactor CT3 for Consider Cranial and Spine
- SeeFactor CT3 for Consider Lung Imaging
- BioScan Touch i8 for Clinical Application - Cardiology
- BioScan Touch i8 for Clinical Application - Renal
- BioScan Touch i8 for Clinical Application - ICU Fluid Balance
- BioScan Touch i8 for Clinical Application - Oncology
- BioScan Touch i8 for Clinical Application - Malnutrition
Degenerative Disease Equipment & Supplies
79 equipment items found
Manufactured by:Neo Medical Sa based inVillette, SWITZERLAND
As we age, the water and protein content of the body’s cartilage changes. This change results in weaker, thinner, and more fragile cartilage. Because both the discs and the joints that connecting the vertebrae (facet joints) are partly composed of cartilage, these areas are subject to degenerative changes. These changes causing the disc to herniate, bulge, or leak. The bulges or leakages ...
by:Gelmetix Limited based inCheshire, UNITED KINGDOM
A focus on low-back pain. About 40% of chronic low-back pain is the result of degenerative disc disease3. Gelmetix’s double cross-linked microgel (DXM) is a biological solution that addresses the source of pain. In the intervertebral disc it restores water content and helps cells to return to normal function1. Our product is not a ...
Manufactured by:K.K. Prosthetic & Orthopaedic Equipment Sdn. Bhd. based inKota Kinabalu, MALAYSIA
Cervical tendinitis., 2. Contractual diathesis., 3. Neck Pain Syndrome., 4. Osteoarthritis., 5. Ligament-muscle lesion., 6. Degenerative ...
Manufactured by:ACS Biotech based inVilleurbanne, FRANCE
Cartilage defects can result either from an accident (car, sport ...) or from a degenerative disease such as osteoarthritis, rheumatoid arthritis or ...
Manufactured by:Stemedica Cell Technologies, Inc. based inSan Diego, CALIFORNIA (USA)
There are two types of cells currently used for investigating progenitor cell therapies for degenerative diseases: autologous and allogeneic. Autologous progenitor cells are derived from the patient being treated. They are retrieved surgically, expanded, and transplanted back into the same patient. Allogeneic progenitor cells are derived from healthy volunteers ...
Manufactured by:BRIM Biotechnology, Inc. based inTaipei City, TAIWAN
Neurotrophic Keratitis is a rare, degenerative disease characterised by corneal sensitivity reduction, spontaneous epithelium breakdown, and impairment of corneal healing. Currently, there is only one drug, Oxervate (Cenegermin), approved by the US FDA and EMA in 2018. Eye health is crucial to quality of life and BRIM is advancing new therapeutics to support ...
Manufactured by:Trulife based inTallaght, IRELAND
C3-C6, ligament/muscular injuries, degenerative joint disease, heumatoid ...
by:Aurora Spine, Inc. based inToronto, ONTARIO (CANADA)
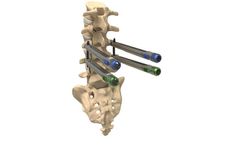
The ZIP ULTRA implant is designed for stabilization and load sharing in T1-S1 thoracolumbar fusion procedures, specifically for the treatment of degenerative disease, lumbar spinal stenosis, spondylolisthesis, trauma, and/or tumor. The proprietary ZIP ONE-STEP™ locking mechanism eliminates the use of a set screw. Each ZIP ULTRA implant features a large ...
Manufactured by:ChoiceSpine LLC based inKnoxville, TENNESSEE (USA)
The ChoiceSpine THUNDERBOLT™ Minimally Invasive Pedicle Screw System is intended to provide immobilization and stabilization of spinal segments in skeletally mature patients as an adjunct to fusion for degenerative disc disease, spondylolisthesis, trauma, spinal stenosis, deformities, tumor, and/or ...
Manufactured by:IPG Photonics Corporation based inMarlborough, MASSACHUSETTS (USA)
Over more than five decades, use of lasers in ophthalmology has successfully shown effective and safe results in treating various eye conditions. Whether lasers are used to correct vision or repair damage due to degenerative diseases dictates optimal laser type, wavelength, and pulse length. ...
Manufactured by:Bioplate based inPlacentia, CALIFORNIA (USA)
Indicated for replacing bony voids in cranial and craniofacial skeletons, excluding maxillofacial regions, these implants avoid complications such as infection and poor blood supply, enhancing the reliability in surgical outcomes. It's crucial to avoid use in patients with systemic infections or significant degenerative diseases. Potential risks include material ...
Manufactured by:MedChemExpress LLC (MCE) based inMonmouth Junction, NEW JERSEY (USA)
N-Nitroso-N-methylurea is a direct-acting alkylating agent that interacts with DNA. N-Nitroso-N-methylurea targets multiple animal organs to cause various cancer and/or degenerative disease. N-Nitroso-N-methylurea is also a precursor in the synthesis of ...
Manufactured by:MedChemExpress LLC (MCE) based inMonmouth Junction, NEW JERSEY (USA)
DAPT also induces autophagy and apoptosis. DAPT has neuroprotection activity and has the potential for autoimmune and lymphoproliferative diseases, degenerative disease and cancers ...
Manufactured by:Xtant Medical based inBelgrade, MONTANA (USA)
It is intended for spinal fusion procedures at one level (C3–T1 inclusive) in skeletally mature patients for the treatment of degenerative disc disease. ...
Manufactured by:Spineway S.A based inÉcully, FRANCE
The posterior fixation techniques stabilize and immobilize the thoracic, lumbar, sacral, and iliac spine using anchorage implants such as pedicle screws and sacral plates. These systems provide treatment options for a range of conditions, including degenerative diseases and complex spinal deformities. Designed and manufactured in France, Spineway's systems are ...
Manufactured by:Excite Medical Corp. based inTampa, FLORIDA (USA)
As with the DRX9000®, the DRX9500® enables healthcare providers to treat patients neck pain caused by herniated discs, bulging discs, and degenerative disc disease. The DRX9500® is offered in a few select clinics that also offer the DRX9000® non-surgical spinal decompression treatment. To find the neared DRX9500® or DRX9000® locations near ...
Manufactured by:Trulife based inTallaght, IRELAND
Manufactured with non-ferrous material, Used to support and stabilize cervical spine and upper thoracic spine after injury or surgery. C2-T3 stable fractures, muscular/ligamentous injury, degenerative joint disease, cervical pain/nerve impingement, Contraindications: Unstable fractures, type II dens fractures ...
Manufactured by:Xtant Medical based inBelgrade, MONTANA (USA)
The Axle Interspinous Fusion System is designed to provide spinal stability for lumbar fusion procedures, including the treatment of degenerative disc disease, spinal tumors and trauma. ...
Manufactured by:ChoiceSpine LLC based inKnoxville, TENNESSEE (USA)
ChoiceSpine’s TigerShark™ C is revolutionizing spinal surgery for patients with degenerative disc disease (DDD). The Anterior Cervical Spacer System utilizes additively manufactured titanium to overcome limitations associated with PEEK and machine titanium. The intervertebral body fusion devices are stronger than other titanium implants, creating ...
Manufactured by:MedChemExpress LLC (MCE) based inMonmouth Junction, NEW JERSEY (USA)
Mavacoxib is a selective, oral long-acting cyclooxygenase-2 (COX-2) inhibitor and a long-acting non-steroidal anti-inflammatory drug (NSAID). Mavacoxib is used to treat pain and inflammation associated with degenerative joint disease in ...