- Home
- Equipment
- micronesia
- in vitro test
Show results for
Refine by
In Vitro Test Equipment & Supplies In Micronesia
104 equipment items found
Manufactured by:ORGENTEC Diagnostika GmbH is part of the Sebia Group based inMainz, GERMANY
AMA-M2 is an ELISA-based, automated, in-vitro test system for the quantitative determination of IgG antibodies against mitochondrial M2 subtype antigen in human serum or plasma. Anti-mitochondrial antibodies (AMA) are a heterogeneous group of autoantibodies directed against various proteins of the outer and inner mitochondrial membrane. ...
Manufactured by:ORGENTEC Diagnostika GmbH is part of the Sebia Group based inMainz, GERMANY
Anti-DGP IgA is an ELISA-based, automated, in-vitro test system for the quantitative determination of IgA antibodies against deamidated gliadin protein epitopes (DGP) in human serum or plasma. These assays are used to support a diagnosis of celiac disease. ...
Manufactured by:ORGENTEC Diagnostika GmbH is part of the Sebia Group based inMainz, GERMANY
Anti-Cathepsin G is an ELISA-based, automated, in-vitro test system for the quantitative determination of IgG antibodies against cathepsin G in human serum or plasma. It is used for the differentiation of a positive pANCA immunofluorescence result. Cathepsin G antibodies are found in cases of systemic vasculitis, inflammatory rheumatic diseases (SLE, ...
Manufactured by:ORGENTEC Diagnostika GmbH is part of the Sebia Group based inMainz, GERMANY
Anti-Insulin is an ELISA-based, automated, in-vitro test system for the quantitative determination of IgG antibodies against animal (beef, pork) and human insulin in human serum or plasma. Anti-Insulin is an ELISA test for the quantitative determination of IgG antibodies against human, bovine and pig insulin in human serum or plasma. ...
Manufactured by:ORGENTEC Diagnostika GmbH is part of the Sebia Group based inMainz, GERMANY
Anti-TG is an ELISA-based, automated, in-vitro test system for the quantitative determination of IgG autoantibodies against thyroglobulin (TG) in human serum or plasma. The protein thyroglobulin is responsible for the production and storage of thyroid hormones and is a strong autoantigen. ...
Manufactured by:ORGENTEC Diagnostika GmbH is part of the Sebia Group based inMainz, GERMANY
Anti-BPI is an ELISA-based, automated, in-vitro test system for the quantitative determination of IgG antibodies against bactericidal permeability-increasing protein BPI in human serum or plasma. Antineutrophil cytoplasmic antibodies (cANCA) against bactericidal permeability-increasing protein (BPI), in addition to PR3 or MPO antibodies, are important serological ...
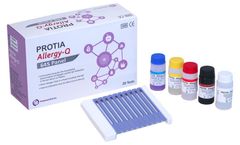
Manufactured by:PROTIA. iNC, formely known as ProteomeTech Inc. based inSeoul, SOUTH KOREA
PROTIA Allergy-Q 64S panel is an in vitro diagnostic test in the quantitative determination of allergen-specific IgE concentrations in human serum or plasma using immunoblotting technique. Diagnose 63 types of allergens including Total IgE in one ...
Manufactured by:Omega Diagnostics Group PLC based inAlva, UNITED KINGDOM
VISITECT CD4 in-vitro diagnostic test is for use as an aid in the management of patients with pre-diagnosed HIV infection. This visually read test is designed to be used at the point-of-care and therefore has utility in decentralised diagnostic settings. The VISITECT® CD4 Rapid Test is a semi-quantitative assay for the ...
Manufactured by:Edif Instruments s.r.l. based inRoma, ITALY
The SARS-CoV-2 IgM ELISA kit is an in vitro diagnostic test for the qualitative detection of IgM antibodies to SARS-CoV-2 in human serum. The kit is intended for use as an aid in identifying individuals with an adaptive immune response to SARSCoV-2, indicating recent or prior infection. ...
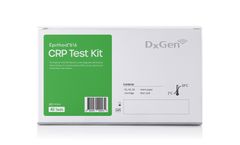
Manufactured by:DxGen Corp. based inGunpo-si, SOUTH KOREA
Test Kit is for in-vitro diagnostic (IVD) use to determine C-reactive protein (CRP) quantitatively in serum/plasma, or whole blood from a human. It provides information for the diagnosis, therapy, and monitoring of inflammatory ...
Manufactured by:CLONIT srl based inAbbiategrasso, ITALY
DuplicαRealTime Next Factor V H1299R Kit is an in vitro diagnostic test for the detection of the nucleotide substitution A4070G of the blood coagulation Factor V gene, by Real-Time ...
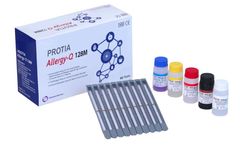
Manufactured by:PROTIA. iNC, formely known as ProteomeTech Inc. based inSeoul, SOUTH KOREA
PROTIA Allergy-Q 128M panel is an in vitro diagnostic test in the quantitative determination of allergen-specific IgE concentrations in human serum or plasma using immunoblotting technique. Diagnose 118 types of allergens including Total IgE in one ...
Manufactured by:Firalis Group based inHuningue, FRANCE
NEUROLINCS : A non-invasive in-vitro diagnostic test measuring 7494 brain-enriched long non-coding RNAs (lncRNAs) that are specific to neurodegenerative ...
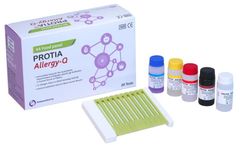
Manufactured by:PROTIA. iNC, formely known as ProteomeTech Inc. based inSeoul, SOUTH KOREA
PROTIA Allergy-Q 64 Food panel is an in vitro diagnostic test in the quantitative determination of allergen-specific IgE concentrations in human serum or plasma using immunoblotting technique. Diagnose 72 types of food allergens including Total IgE in one ...
Manufactured by:PROTIA. iNC, formely known as ProteomeTech Inc. based inSeoul, SOUTH KOREA
PROTIA Food IgG a is an in vitro diagnostic test with the semi-quantitative determination of food specific IgG (sIgG) in human serum using immunoblotting method. Diagnose 60 types of allergens (inducing food intolerance) in one ...
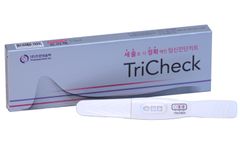
Manufactured by:PROTIA. iNC, formely known as ProteomeTech Inc. based inSeoul, SOUTH KOREA
TriCheck, an in vitro self-testing kit, uses immunochromatography method for the qualitative test of hCG (human Chorionic Gonadotropin) in human urine samples to help the determination of ...
Manufactured by:Coris BioConcept based inGembloux, BELGIUM
In vitro rapid diagnostic test for the detection of SARS-CoV-2 antigen in nasopharyngeal secretions. Since many months, several variants did pop-up from countries where the pandemic was particularly active (i.e. UK, Brazil, India and South Africa) and our test has been proven to detect all of ...
Manufactured by:Abionic SA based inEpalinges, SWITZERLAND
The IVD CAPSULE Aeroallergens is the first rapid, single-use in vitro diagnostic test for the quantitative determination of circulating immunoglobulin E (IgE) specific to the allergen components Phl p 1 + Phl p 5 (combined), Der p 1 + Der p 2 (combined), Alt a 1, Fel d 1 and Can f 1 in uncoagulated human capillary blood collected from the patient’s ...
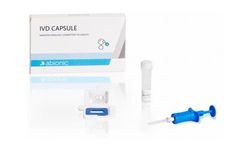
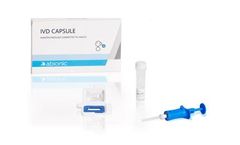
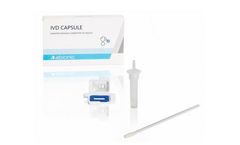
Manufactured by:Abionic SA based inEpalinges, SWITZERLAND
The IVD CAPSULE PSP is a rapid, single-use in vitro diagnostic test for the determination of Pancreatic Stone Protein (PSP) in blood for the calculation of the cSOFA score at the point of care. The cSOFA score is used to assess the severity of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in ...
Manufactured by:Abionic SA based inEpalinges, SWITZERLAND
The IVD CAPSULE COVID-19 is a rapid, single-use in vitro diagnostic test that measures the presence of a specific SARS-CoV-2 viral antigen in saliva. Abionic’s COVID-19 saliva test allows for ultra-fast screening of COVID-19 patients at the point-of-care with lab quality ...