Show results for
Refine by
Patient Management Equipment Supplied In Canada
10 equipment items found
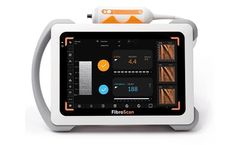
Manufactured by:Echosens based inParis, FRANCE
The mobile non-invasive solution for liver disease management, ideal for multi-site configuration. Powered by LSM by VCTE™ and CAP™ for liver fibrosis and liver steatosis assessment. FibroScan® Mini+ 430 is designed for multi-center sharing and remote liver patient ...
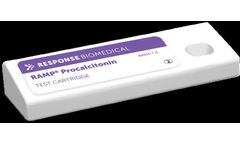
Manufactured by:Response Biomedical Corp. based inVancouver, BRITISH COLUMBIA (CANADA)
Procalcitonin (PCT) is a biomarker elevated in the blood of patients suffering from sepsis. Physicians use PCT to distinguish bacterial sepsis from other causes of similar symptoms. RAMP Procalcitonin provides quantitative results in ~15 minutes directly from a small blood sample. When speed, precision and accuracy are critical, the RAMP Procalcitonin test provides clinicians ...
Manufactured by:Augurex Life Sciences Corp. based inVancouver, BRITISH COLUMBIA (CANADA)
The 14-3-3η blood test is clinically available as a diagnostic test whereby a positive result indicates a 5 to 50 times greater likelihood of having RA and informs joint damage prognosis and disease monitoring. 14-3-3η is used in combination with other serological tests and clinical assessment to support RA patient ...
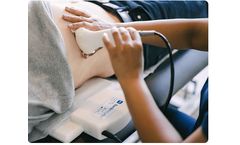
Manufactured by:Sonic Incytes Medical Corp. based inVancouver, BRITISH COLUMBIA (CANADA)
It generates real-time stiffness measurements at point of care with technology similar to that of MRI elastography. Velacur is safe and effective as an aid in the management of patients with liver disease. ...
Manufactured by:OraSure Technologies based inBethlehem, PENNSYLVANIA (USA)
Testing for Ebola Virus Disease (EVD) must be performed in accordance with current guidelines provided by the appropriate public health authorities that address appropriate biosafety conditions, interpretation of test results, and coordination of testing, results and patient management with public health authorities. ...
Manufactured by:OraSure Technologies based inBethlehem, PENNSYLVANIA (USA)
Testing for Ebola Virus Disease (EVD) must be performed in accordance with current guidelines provided by the appropriate public health authorities that address appropriate biosafety conditions, interpretation of test results, and coordination of testing, results and patient management with public health ...
Manufactured by:Beckman Coulter, Inc. based inBrea, CALIFORNIA (USA)
The iRICELL series combines urine chemistry and microscopy in a fully automated, walk-away workcell that is easy to use and maintain. Optimize and advance urinalysis and body fluid testing in your laboratory with proprietary Digital Flow Morphology technology that uses Auto-particle Recognition (APR) software to deliver standardized and accurate ...
Manufactured by:Echosens based inParis, FRANCE
The state-of-the-art non-invasive solution, affordable to all. Powered by LSM by VCTE™ and CAP™ for liver fibrosis and steatosis management. Supported by 6 270+ peer reviewed publications and 250+ guidelines. An ideal solution for point-of-care use: Pay-per-exam solution. A full ecosystem of solutions,including service coverage and support. Centralized data management and ...
Manufactured by:Beckman Coulter, Inc. based inBrea, CALIFORNIA (USA)
The iQ200 series automated urine microscopy analyzer produces shortened TAT with standardized results. By leveraging proprietary Digital Flow Morphology technology using Auto-Particle Recognition (APR) Software, urine particles are isolated, identified and characterized on the screen to virtually eliminate the need for manual microscopy. The iQ200 series analyzers are available as either ...
Manufactured by:Response Biomedical Corp. based inVancouver, BRITISH COLUMBIA (CANADA)
Response Biomedical offers a FDA cleared whole blood immunoassay NT-proBNP test, with our proprietary RAMP technology which detects the levels of N-terminal prohormone of brain natriuretic peptide in patients with congestive heart failure around the world. Read more about how this product will help your physicians and patients where a traditional BNP test ...