Refine by
Applications
- Medical Lasers Systems for Laser Treatments
- Medical Lasers Systems for Dental Lasers
- Medical Lasers Systems for Podiatry Lasers
- Medical Lasers Systems for Laser Acupuncture
- Medical Lasers Systems for Chiropractic Lasers
- Medical Lasers Systems for Physical Medicine
- Medical Lasers Systems for Laser Massage Therapy
- Medical Lasers Systems for Laser Physical Therapy
- Actionable Molecular Diagnostic System for Physicians
Patient Selection Equipment & Supplies
113 equipment items found
Manufactured by:Pulmonx Corporation based inRedwood City, CALIFORNIA (USA)
The Zephyr Endobronchial Valve is an implantable device used to occlude all airways feeding the hyperinflated lobe of a lung that is most diseased with ...
Manufactured by:Kura Oncology, Inc. based inSan Diego, CALIFORNIA (USA)
With the advent of precision medicines that can target unique molecular or genetic features of a patient’s tumor, the field of oncology has a host of alternatives to one-size-fits-all treatments. This is a major advance over traditional chemotherapy, which can be as potent against healthy tissue as it is against cancer. Using tumor genomic profiling and biomarker ...
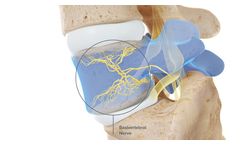
Manufactured by:Relievant Medsystems, Inc. based inMinneapolis, MINNESOTA (USA)
The Intracept Procedure for the Relief of Chronic Vertebrogenic Low Back Pain. The Intracept Procedure is a minimally invasive procedure that targets the basivertebral nerve for the relief of chronic vertebrogenic low back ...
by:Oxford Brain Diagnostics Ltd. based inOxford, UNITED KINGDOM
Using CDM® we will optimise patient selection, combine other clinical and patient demographics, and positively impact trial costs and performance. CDM® can estimate a patient’s pathology disease stage using grey matter quality. We provide a likelihood that a patient will progress to the ...
Manufactured by:CytomX Therapeutics, Inc. based inSouth San Francisco, CALIFORNIA (USA)
Bristol Myers Squibb is enrolling patients in the dose escalation phase of a Phase 1/2a clinical trial (NCT03994601) of a second anti-CTLA-4 Probody, BMS-986288, based on a modified version of Yervoy® (ipilimumab), to evaluate a CTLA-4-directed Probody therapeutic alone or in combination with Opdivo® (nivolumab) in patients with selected ...
Manufactured by:Medoc based inRamat Yishay, ISRAEL
The challenge in performing pressure algometry tests is to apply continuously ascending pressure at a constant rate on the patient's selected body site without having the possibility to monitor it in real time. ...
Manufactured by:Summit Industries, LLC based inNiles, ILLINOIS (USA)
Features: Anatomical Programming, Pedestal Mount, DR Integration, available, Constant Potential Generator maximizes image quality and consistency, Designed for reliability and low cost of ownership, Anatomical Programming with six selectable patient sizes ensures flexibility in choosing the right techniques for your patient exam, Dual Speed ...
Manufactured by:Aposcience AG based inVienna, AUSTRIA
Aposcience AG will initiate a program to demonstrate safety and efficacy in this important indication with very large unmet medical need. The program consists of a phase I study in healthy volunteers, followed by a phase IIa study in a selected patient population. This program is designed to translate the extremely promising data from animal models into clinical ...
Manufactured by:Organovo Holdings Inc. based inSan Diego, CALIFORNIA (USA)
While the bioprinting process is cell-agnostic, our bioprinted tissues utilize primary human cells where possible in order to produce the in vivo-like physiology most relevant for drug testing. We utilize multiple independent sources for our input cells to allow patient selection and high levels of ...
Manufactured by:MiraDx based inLos Angeles, CALIFORNIA (USA)
MiraDx discovered a panel of such mutations that predict both response and toxicity to anti-PD1 and anti-PDL1 therapies currently on the market. We are working with potential partners to incorporate these findings into the newer agents to allow better patients selection to achieve improved response and less toxicity from these exciting new therapies. We are ...
Manufactured by:Senzime AB Uppsala based inUppsala, SWEDEN
Device connectivity- data from the TetraGraph Monitor can be transferred to select patient monitoring systems, these NMT data can be integrated into existing infrastructure, allowing the uploading of patient data into the local electronic medical record ...
Manufactured by:OrthoPediatrics Corp. based inWarsaw, INDIANA (USA)
Designed to fill a crucial gap in the treatment of adolescent idiopathic scoliosis (AIS), the ApiFix procedure provides a viable alternative to both bracing and fusion for select patients. Less Invasive: With three pedicle screws and a familiar posterior approach, the unilateral technique is less invasive than spinal fusion and simpler than the other fusionless ...
Manufactured by:Teleflex Incorporated based inMorrisville, NORTH CAROLINA (USA)
The EZ-Blocker Endobronchial Blocker is the next generation of one-lung ventilation products designed to enhance patient outcomes during selective ...
Manufactured by:Dermagnostix GmbH based inFreiburg, GERMANY
PatternX identifies inflammatory disease markers, characterizes endotypes, and supports companion and complementary diagnostics development, as well as patient stratification and selection in clinical ...
Manufactured by:EndoGastric Solutions, Inc. based inRedmond, WASHINGTON (USA)
Healthcare professionals should be consulted for all risks and benefits of all treatment options. Trained physicians are familiar with patient selection criteria most associated with positive clinical outcomes and can determine whether the TIF procedure is appropriate on a case-by-case basis. While clinical studies support the effectiveness of TIF (Transoral ...
Manufactured by:ApiFix Ltd. based inYokneam Illit, ISRAEL
Bracing may be ineffective. Fusion is forever. The ApiFix® procedure offers select patients with adolescent idiopathic scoliosis (AIS) a less invasive, motion-preserving option for achieving curve correction – without the permanence of ...
Manufactured by:GENCURIX based inGuro-gu, SOUTH KOREA
These tests detect DNA mutations by analyzing certain genes extracted from the patient's blood and tumor samples based on ddPCR platform which is optimized for companion diagnostics. They select patients who benefit the effectiveness of cancer ...
Manufactured by:Alphageneron based inCambridge, MASSACHUSETTS (USA)
An allogenic NK cell therapy that targets tumors expressing mHsp70. Patients are selected with high mHsp70 exosomes in their blood using our Companion Diagnostic AP-CDx (described below). We obtain PBMCs from healthy donors by apheresis, their NK cells are expanded, and incubated ex vivo with TKD and IL-2, followed by freezing, thawing and intravenous ...
Manufactured by:Berlin Heart GmbH based inBerlin, GERMANY
It is indicated for children with life-threatening heart failure after all conservative therapeutic options have been exhausted. EXCOR® Pediatric is designed for use in young patients, from newborns to adolescents. Berlin Heart offers a comprehensive service plan for this system. Our Clinical Affairs Team supports you for the entire duration of VAD therapy: starting with ...
Manufactured by:Swissray Medical AG based inHochdorf, SWITZERLAND
The completely motorized ddRAura U is quickly and effortlessly positioned with Swissray’s APS – Automatic positioning system via handheld remote control, optional wireless foot pedals or ddRAura workstation control. The ddRAura’s tube mounted touchscreen provides control generator, collimator, system position, patient procedure list selection ...