Refine by
Locations
- USA
- Alabama
- Alaska
- Arizona
- Arkansas
- California
- Colorado
- Connecticut
- Delaware
- District Of Columbia
- Florida
- Georgia (USA)
- Hawaii
- Idaho
- Illinois
- Indiana
- Iowa
- Kansas
- Kentucky
- Louisiana
- Maine
- Maryland
- Massachusetts
- Michigan
- Minnesota
- Mississippi
- Missouri
- Montana
- Nebraska
- Nevada
- New Hampshire
- New Jersey
- New Mexico
- New York
- North Carolina
- North Dakota
- Ohio
- Oklahoma
- Oregon
- Pennsylvania
- Rhode Island
- South Carolina
- South Dakota
- Tennessee
- Texas
- Utah
- Vermont
- Virginia
- Washington
- West Virginia
- Wisconsin
- Wyoming
Cortical Bone Equipment Supplied In USA
52 equipment items found
Manufactured by:ATEC Spine, Inc based inCarlsbad, CALIFORNIA (USA)
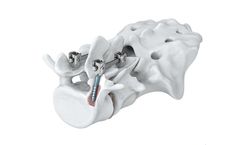
The Arsenal Cortical Bone Fixation (CBx) System is designed to provide instrumentation and implants for a medial to lateral pedicular approach for spinal fixation. The CBx System is built off of the Arsenal Spinal Fixation Platform and features many of the same benefits, instruments, markings, color coding, and layout as the Arsenal Spinal Fixation ...
Manufactured by:Xtant Medical based inBelgrade, MONTANA (USA)
Xtant’s 3Demin Technology utilizes demineralized cortical bone fibers that are entangled and shaped into sizes engineered to complement specific surgical applications. This unique process creates an interconnected graft material that contains BMPs and other growth factors necessary for the promotion of new bone formation. 3Demin allografts ...
Manufactured by:Isto Biologics based inHopkinton, MASSACHUSETTS (USA)
An advanced allograft that combines the regenerative capacity of interconnected fibers with the availability of growth factors endogenous to cortical ...
Manufactured by:Impladent Ltd. based inJamaica, NEW YORK (USA)
This product contains Human Allograft Material (referred to as “Graft”) collected from human cadaveric donors. All tissue processed for Impladent Ltd. is recovered by U.S. tissue banks as well as a completed donor chart for the enclosed product including but not limited to: serology results, preprocessing culture results, medical and social history evaluation and serodilution ...
Manufactured by:Maxtown Medical based inWesterville, OHIO (USA)
MX T2111 cases.. 81-1461:- forward straight, 1-0. 81-1462:- forward straight, 2-0. 81-1463:- forward straight, 3-0. 81-1464:- forward straight, 4-0. 81-1465:- forward straight cortical bone cutter. 81-1466:- forward angle, 1-0. 81-1467:- forward angle, 2-0. 81-1468:- forward angle, 3-0. 81-1469:- forward angle, 4-0. 81-1470:- forward angle ...
Manufactured by:VIVEX Biologics, Inc. based inMiami, FLORIDA (USA)
VIA Fill™ was developed utilizing VIVEX’s proprietary Integrity Processing™, where cortical bone fibers are demineralized, exposing the natural bone morphogenic proteins (BMPs) needed for bone formation.1 VIA Fill™ is moldable and comprised of 100% demineralized cortical ...
Manufactured by:Ortho-Medical GmbH based inDuerbheim, GERMANY
ORLOS_II LOckING PLATE SYSTEM 4,5 - 5.0 MM • Angular stable • Locking or compression • Support biological osteosynthesis in case of osteoporotic bone and multiple fractures • Excessive torque is not applied to the cortical bone • Self-tapping locking screws • The bonefragments are are reliably fi xed in the position ...
Manufactured by:VIVEX Biologics, Inc. based inMiami, FLORIDA (USA)
VIA DBM Plus™ is a demineralized bone matrix putty containing cortical bone chips and indicated for use in the extremities, posterolateral spine, and pelvis as a bone void filler or for treatment of osseous ...
Manufactured by:Berkeley Advanced Biomaterials (BAB) based inBerkeley, CALIFORNIA (USA)
Demineralized Bone Matrix (DBM) is produced from ground cortical bone and contains osteoinductive proteins. It is intended to be used to fill bone defects and ...
Manufactured by:ExsoMed Corporation based inAliso Viejo, CALIFORNIA (USA)
InFrame is designed specifically for phalanx fractures to provide surgeons with a reliable solution through a simple, minimally invasive approach. The robust length offering is intended to accurately fit the intramedullary canal and achieve bi-cortical bone purchase, creating stable fixation and precise reduction for all types of phalanx ...
Manufactured by:Aziyo Biologics, Inc. based inSilver Spring, MARYLAND (USA)
ViBone is comprised of cancellous bone particles with preserved cells and demineralized cortical bone particles produced via Aziyo’s gentle proprietary VBM ...
Manufactured by:Spine Wave, Inc. based inShelton, CONNECTICUT (USA)
Tempest® Allograft Bone Matrix is a sterile, ready-to-use allograft bone matrix composed of cancellous and partially demineralized cortical bone for use in filling a variety of bone voids. Its proprietary processing yields a bone allograft product with unique handling characteristics that is ...
Manufactured by:Orthofix Medical Inc. based inLewisville, TEXAS (USA)
The Trinity Evolution allograft is comprised of cancellous bone with viable osteogenic and osteoprogenitor cells retained within the matrix and a demineralized cortical bone component. The Trinity Evolution allograft offers an ideal alternative to autograft and other bone grafting ...
Manufactured by:Cervos Medical based inMarshfield, MASSACHUSETTS (USA)
The CER-EXT-M has a profile that makes it ideal for use in extremity bones. The enhanced features of the tip of the stylet and cannula facilitate penetrating harder cortical bone including the distal tibia. Its shorter length makes it ergonomically friendly for working in extremity locations and it incorporates patented technology that limits ...
Manufactured by:Microport Orthopedics Inc. based inArlington, TENNESSEE (USA)
The Profemur® line of primary hip stems has an established clinical history. Each Profemur® hip stem offers its own philosophy to provide fixation and replace bone anatomy. Triple taper stem philosophy" Designed for primary hip replacement surgery, the Profemur® Gladiator® hip stem is a triple tapered wedge stem with a distinct geometry. The hip stem is designed ...
Manufactured by:OrthoProMed Inc. based inCovina, CALIFORNIA (USA)
The PEEK Interference Screw by Orthopromed is designed for reliable mechanical fixation in ACL and PCL reconstruction procedures. It addresses both bone-patellar tendon-bone (BTB) and soft tissue grafts, featuring a thread pattern that facilitates a straightforward surgical technique requiring minimal tunnel preparation. PEEK, or polyether ether ketone, is chosen ...
Manufactured by:Xtant Medical based inBelgrade, MONTANA (USA)
Osteogenic Potential- Proprietary processing preserves adult mesenchymal stem cells while removing red and white blood cells. Osteoinductive Potential- Cortical fiber demineralized bone matrix provides a high quality osteoinductive signal. Additional growth factors are captured from bone lining cells and restored back into the product.* ...
Manufactured by:Onkos Surgical based inParsippany, NEW JERSEY (USA)
Via Graft provides cells and a three dimensional scaffold necessary for bone repair. The scaffold is comprised of a proprietary blend of microparticulate cortical, cancellous and demineralized cortical allograft bone. Optimal particle sizes range from 100-300 microns. Via Graft processing preserves a cell population that includes ...
Manufactured by:Onkos Surgical based inParsippany, NEW JERSEY (USA)
Via Graft provides cells and a three dimensional scaffold necessary for bone repair. The scaffold is comprised of a proprietary blend of microparticulate cortical, cancellous and demineralized cortical allograft bone. Optimal particle sizes range from 100-300 microns. Via Graft processing preserves a cell population that includes ...
Manufactured by:Nanova Biomaterials, Inc. based inColumbia, MISSOURI (USA)
FiberFIX™ Screw-in Anchor is preloaded with 2 #2 sutures for soft tissue repair. The screw is fully threaded to secure engagement throughout the bone, both cortical and cancellous. The combination of nanofiber reinforced composite and our unique design delivers optimal insertion with high max torque and pullout strength for repair ...