Show results for
Refine by
Locations
- USA
- Alabama
- Alaska
- Arizona
- Arkansas
- California
- Colorado
- Connecticut
- Delaware
- District Of Columbia
- Florida
- Georgia (USA)
- Hawaii
- Idaho
- Illinois
- Indiana
- Iowa
- Kansas
- Kentucky
- Louisiana
- Maine
- Maryland
- Massachusetts
- Michigan
- Minnesota
- Mississippi
- Missouri
- Montana
- Nebraska
- Nevada
- New Hampshire
- New Jersey
- New Mexico
- New York
- North Carolina
- North Dakota
- Ohio
- Oklahoma
- Oregon
- Pennsylvania
- Rhode Island
- South Carolina
- South Dakota
- Tennessee
- Texas
- Utah
- Vermont
- Virginia
- Washington
- West Virginia
- Wisconsin
- Wyoming
Healing Improvement Equipment Supplied In USA
93 equipment items found
Manufactured by:Links Medical Products Inc. based inIrvine, CALIFORNIA (USA)
Our Manuka honey is fine filtered, rigorously tested and sterilized, making it appropriate for application to most wounds. Available in 1.5oz and 0.5oz tubes. Manuka Fill helps maintain a moist healing environment conducive to improving wound healing. ...
Manufactured by:Suneva Medical based inSan Diego, CALIFORNIA (USA)
Extreme device precision and controlled energy settings limit the damage of surrounding skin, reduce downtime and improve healing. Plasma IQ treatment can be performed in-office in as little as 30 minutes, depending on the area(s) treated. Two levels of energy allow for safe and effective treatments for a variety of indications and areas. Plasma IQ delivers the ...
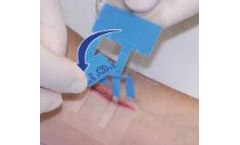
Manufactured by:Clozex Medical, Inc. based inWatertown, MASSACHUSETTS (USA)
Interlaced Locking Technology aligns and holds skin edges at the same height for optimal healing and great cosmetic ...
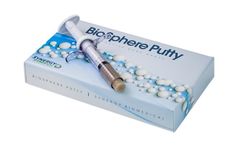
Manufactured by:Synergy Biomedical, LLC based inWayne, PENNSYLVANIA (USA)
The combination of the BioSphere particles and a resorbable, phospholipid carrier results in a bioactive Putty with the highest bioactive glass content on the market, excellent handling, and improved bone ...
Manufactured by:Rumex International Co. based inTampa, FLORIDA (USA)
Designed for making a paracentesis (to insert a subordinate instrument) Titanium handle with locking mechanism. Extreme sharpness of the blades. Minimal pressure. Up to 3000 incisions. Natural diamond. Predictable cuts and architecture of the wound for more consistent healing. Potentially reduced risk of wound leak. The cutting edge of 4-10 nm pushes the molecules apart without damaging them and ...
Manufactured by:Rumex International Co. based inTampa, FLORIDA (USA)
Designed for making a paracentesis (to insert a subordinate instrument) Titanium handle with locking mechanism. Extreme sharpness of the blades. Minimal pressure. Up to 3000 incisions. Natural diamond. Predictable cuts and architecture of the wound for more consistent healing. Potentially reduced risk of wound leak. The cutting edge of 4-10 nm pushes the molecules apart without damaging them and ...
Manufactured by:InHealth Technologies based inCarpinteria, CALIFORNIA (USA)
Gentle and flexible while still attaching firmly to the skin. Strong adhesion yet easily removable. Absorbs sweat while still maintaining adhesion. Moist environment for optimized healing. ...
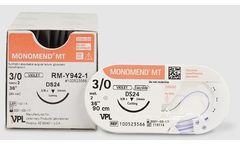
Manufactured by:PRN Pharmacal based inPensacola, FLORIDA (USA)
With unparalleled needle sharpness and monofilament options for all indications VPL sutures provide smooth passage through tissue improving would healing times and post-surgical comfort. Monomend® MT is a violet, monofilament mid-term, synthetic absorbable suture providing 14 days of wound support with complete absorption in 60-90 days (0% initial tensile ...
Manufactured by:Advanced Oxygen Therapy Inc. based inOceanside, CALIFORNIA (USA)
The TWO2 therapy Multi-Patch system is comprised of two major components: A single use Multi-Patch chamber and an oxygen source. An oxygen flow rate of ≈ 4 lpm applied into the chamber delivers 30 mb of non-contact pressure directly to the wound site, within a sealed and humidified environment. The resultant oxygen partial pressure diffusion gradient of close to 800 mmHg provides for ...
Manufactured by:Acera Surgical Inc. based inLouis, MISSOURI (USA)
For clinicians treating patients with partial-thickness burns, Covora is a non-biologic, hybrid-scale fiber matrix, engineered to be similar to human ECM that is strong and pliable, covers sensitive burn areas, and promotes ...
Manufactured by:NAQI nv/sa based inHalen, BELGIUM
Skin regeneration Support of healed skin. Follow up NAQI® Repair: Creates a healthy skin barrier. Promotes skin regeneration. Improves skin elasticity. Improves the appearance and colour of scars. Increases the moisture content of the skin. Reduces flaking. Prevents and reduces wrinkles and striae. ...
Manufactured by:Rumex International Co. based inTampa, FLORIDA (USA)
6-20/6-104 Phaco Diamond Knife, Trapezoid Self-Diving Blade, 2.30/2.80 mm, Length 130 mm, Angled Titanium Handle. Used to perform the main incision/tunnel (for the introduction of a phaco needle). Symmetric sharp edges of the blade ensure even diving inside the tissue with less corneal distortion. Profile of the incision is directed to the anterior chamber. Titanium handle with locking mechanism. ...
Manufactured by:Rumex International Co. based inTampa, FLORIDA (USA)
6-20/6-107 Phaco Diamond Knife, Trapezoid Self-Diving Blade, 2.00/2.30 mm, Angled, Length 130 mm, Titanium Handle. Used to perform the main incision/tunnel (for the introduction of a phaco needle). Symmetric sharp edges of the blade ensure even diving inside the tissue with less corneal distortion. Profile of the incision is directed to the anterior chamber. Titanium handle with locking ...
Manufactured by:Rumex International Co. based inTampa, FLORIDA (USA)
Universal Three-Step Knife for Cataract and LRI Surgery. Precise, easy and stable calibration mechanism with 3 preset depths of 500, 550 and 600 microns. Full extension for side-port incision. Single footplate for better visualization. Extreme sharpness of the blades. Minimal pressure. Up to 3000 incisions. Natural diamond. Predictable cuts and architecture of the wound for more consistent ...
Manufactured by:Azena Medical, LLC. based inWalnut Creek, CALIFORNIA (USA)
The optional Photobiomodulation (PBM) Adapter further enhances the system’s capabilities. ...
Manufactured by:Cabaletta Bio, Inc. based inPhiladelphia, PENNSYLVANIA (USA)
We received Orphan Drug Designation for the treatment of PV from the FDA in January 2020 and Fast Track Designation for improving healing of mucosal blisters in patients with mPV from the FDA in May 2020. The DesCAARTes™ trial is actively recruiting patients at multiple clinical sites across the ...
Manufactured by:Parametrics Medical based inLeander, TEXAS (USA)
Restorigin Sx amniotic membranes are procured through voluntary donation from scheduled cesarean procedures of full-term, live births. Amniotic membranes are applied as a tissue barrier to help provide mechanical protection while supporting healing with endogenous growth factors. Restorigin Sx may be used as a barrier in numerous surgical applications, including but not limited to spine and ...
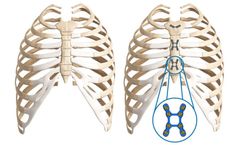
Manufactured by:Zimmer Biomet CMF and Thoracic based inJacksonville, NEW JERSEY (USA)
SternaLock Blu is specifically designed by cardiothoracic surgeons for primary sternal closure following median sternotomies for open-heart procedures. Zimmer Biomet has nearly two decades of clinical experience. Explore the legacy studies and whitepapers of our primary closure ...
Manufactured by:Zimmer Biomet CMF and Thoracic based inJacksonville, NEW JERSEY (USA)
The SternaLock Blu Study was designed by a team of expert cardiothoracic surgeons and leading health economic researchers, and compared outcomes following sternal closure with SternaLock Blu or wire cerclage. A total of 236 patients undergoing elective cardiac surgery were enrolled at 12 hospitals in the United States. Patients were followed for six months to evaluate healing, clinical outcomes ...
Manufactured by:Onkos Surgical based inParsippany, NEW JERSEY (USA)
Cygnus is a regenerative tissue matrix that has innate regenerative capability and supports healing without adhesion or scar formation due to its fetal ...