Show results for
Refine by
Locations
- USA
- Alabama
- Alaska
- Arizona
- Arkansas
- California
- Colorado
- Connecticut
- Delaware
- District Of Columbia
- Florida
- Georgia (USA)
- Hawaii
- Idaho
- Illinois
- Indiana
- Iowa
- Kansas
- Kentucky
- Louisiana
- Maine
- Maryland
- Massachusetts
- Michigan
- Minnesota
- Mississippi
- Missouri
- Montana
- Nebraska
- Nevada
- New Hampshire
- New Jersey
- New Mexico
- New York
- North Carolina
- North Dakota
- Ohio
- Oklahoma
- Oregon
- Pennsylvania
- Rhode Island
- South Carolina
- South Dakota
- Tennessee
- Texas
- Utah
- Vermont
- Virginia
- Washington
- West Virginia
- Wisconsin
- Wyoming
Spinal Surgery Equipment Supplied In USA
111 equipment items found
Manufactured by:Mizuho OSI based inUnion City, CALIFORNIA (USA)
The first of its kind, ProAxis features advanced hinge technology to provide unparalleled surgical access, precision and flexibility. ...
Manufactured by:ChoiceSpine LLC based inKnoxville, TENNESSEE (USA)
ChoiceSpine’s TigerShark™ C is revolutionizing spinal surgery for patients with degenerative disc disease (DDD). The Anterior Cervical Spacer System utilizes additively manufactured titanium to overcome limitations associated with PEEK and machine titanium. The intervertebral body fusion devices are stronger than other titanium implants, creating ...
Manufactured by:Ambler Surgical based inExton, PENNSYLVANIA (USA)
Cone suction cannula, 3 1/2'', straight, 15 gauge, working length ...
Manufactured by:Ad-Tech Medical Instrument Corporation based inOak Creek, WISCONSIN (USA)
For intraoperative monitoring of evoked potential (EPs) of the descending pathway during spinal surgery. Measures ...
Manufactured by:Neurovision Medical Products based inVentura, CALIFORNIA (USA)
DryTouch integrates monopolar stimulation with a Frazier suction instrument for brain mapping, minimally invasive thyroidectomies, and spinal ...
Manufactured by:Mizuho OSI based inUnion City, CALIFORNIA (USA)
With over 8,000 units sold worldwide, the Modular Table System (MTS) delivers precise and stable positioning, with interchangeable tops for spine, imaging and orthopedic trauma procedures. Part of the original modular system, the Spinal Surgery Top offers the classic open frame design for posterior spinal surgery, while the ...
Manufactured by:Xtant Medical based inBelgrade, MONTANA (USA)
The Axle Interspinous Fusion System is an internal fixation device for posterior spinal surgery in the non-cervical spine (T1-S1 inclusive). It is a minimally invasive, modular Interspinous fusion system with straightforward instrumentation. The Axle Interspinous Fusion System is designed to provide spinal stability for lumbar fusion procedures, ...
Manufactured by:Whale Imaging based inWaltham, MASSACHUSETTS (USA)
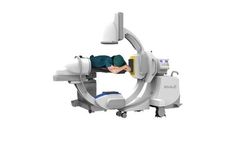
The G-Arm is designed to improve user confidence and patient outcomes in areas such as orthopedics, spinal surgery, neurosurgery, trauma, and pain management. Providing unprecedented capability and control, G-Arm aims to improve your surgical experience. For clinicians and hospitals, Whale’s product development focus is to create potential ...
Manufactured by:Orthofix Medical Inc. based inLewisville, TEXAS (USA)
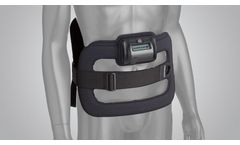
The SpinalStim device is FDA approved to be used after spinal fusion surgery or to be used to treat a failed fusion from a previous surgery. The devices stimulate the natural healing process of bone by sending low-level pulses of electromagnetic energy to the injury or fusion site. The device has an overall clinical success rate of 92% in ...
Manufactured by:Enovis based inLewisville, TEXAS (USA)
CMF Spinalogic is a portable, battery-powered, micro-controlled, noninvasive bone growth stimulator indicated as an adjunct electromagnetic treatment to primary lumbar spinal fusion surgery for one or two ...
Manufactured by:MedSpark, LLC based inSan Luis Obispo, CALIFORNIA (USA)
The pedicle screw, which is sometimes used as an adjunct to spinal fusion surgery, provides a means of gripping a spinal segment. The screws themselves do not fixate the spinal segment, but act as firm anchor points that can then be connected with a rod. The screws are placed by the surgeon into two or three consecutive spine ...
Manufactured by:Ankasa Regenerative Therapeutics based inLa Jolla, CALIFORNIA (USA)
Ankasa is the first to produce human Wnt proteins in a manufacturing setting suitable for use in humans. Our first clinical approach will be the demonstration of improved outcomes after spinal fusion surgeries. Our product has expansion opportunities to other bone reconstructive procedures including osseointegration of dental and orthopedic implants (knee and hip ...
Manufactured by:Shukla Medical based inSt. Petersburg, FLORIDA (USA)
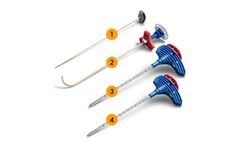
Designed to remove small bone screws commonly used in todays craniomaxillofacial, hand, wrist, foot, ankle, and spine ...
Manufactured by:CoreLink, LLC based inSt. Louis, MISSOURI (USA)
Tiger Deformity provides an extensive breadth of screw, hook, rod, and connector options to address complex spinal conditions and ...
Manufactured by:Shukla Medical based inSt. Petersburg, FLORIDA (USA)
The SHUKLA Copter system is designed to quickly remove entire pedicle screw constructs from the spine, provided the surgeon is able to cut the spinal rod on either side. It is notably useful for all challenging screw constructs where any component (such as a locking cap, set screw, or bone screw) is fixed or the correct driver is not available. ...
Manufactured by:Relievant Medsystems, Inc. based inMinneapolis, MINNESOTA (USA)
The Intracept Access Instruments are engineered for precise targeting of the BVN through a traditional unipedicular approach. The procedure is performed under fluoroscopic imaging, similar to many other minimally invasive spine procedures ...
Manufactured by:Clinicon Corporation based inOceanside, CALIFORNIA (USA)
The PureBeam delivery system is the preferred tool of minimally invasive surgery specialists to treat soft tissue disorders in the field of Neuro-, Spine-, and ENT ...
Manufactured by:OR Specific based inVancouver, WASHINGTON (USA)
Unique absorbent drape easily covers both the upper and main surfaces of the table. The clear plastic backdrop protects the sterile field while allowing light through to the main table surface. Standard: drape provides barrier protection, tear & puncture resistance, and absorbent layer. For OR use. Heavy-Duty: drape provides premium barrier protection, tear & puncture resistance, and high ...
Manufactured by:OR Specific based inVancouver, WASHINGTON (USA)
Only available OUS; Unique absorbent drape easily covers both the upper and main surfaces of the table. The clear plastic backdrop protects the sterile field while allowing light through to the main table surface. Heavy-Duty: drape provides premium barrier protection, tear & puncture resistance, and high capacity absorbent layer. For OR use. Designed specifically for large orthopedic and ...
Manufactured by:Xtant Medical based inBelgrade, MONTANA (USA)
Matriform Si was developed to resemble the composition and porous structure of natural human bone. Comprised of 96% pure phase β-TCP granules, 4% silicate and collagen, Matriform Si provides the ideal biomimetic scaffold for spinal fusion procedures. The flexible strip offers excellent handling and shape memory ensuring direct contact with the surface of healthy ...