Refine by
Locations
- USA
- Alabama
- Alaska
- Arizona
- Arkansas
- California
- Colorado
- Connecticut
- Delaware
- District Of Columbia
- Florida
- Georgia (Us)
- Hawaii
- Idaho
- Illinois
- Indiana
- Iowa
- Kansas
- Kentucky
- Louisiana
- Maine
- Maryland
- Massachusetts
- Michigan
- Minnesota
- Mississippi
- Missouri
- Montana
- Nebraska
- Nevada
- New Hampshire
- New Jersey
- New Mexico
- New York
- North Carolina
- North Dakota
- Ohio
- Oklahoma
- Oregon
- Pennsylvania
- Rhode Island
- South Carolina
- South Dakota
- Tennessee
- Texas
- Utah
- Vermont
- Virginia
- Washington
- West Virginia
- Wisconsin
- Wyoming
Syphilis Equipment Supplied In Usa
29 equipment items found
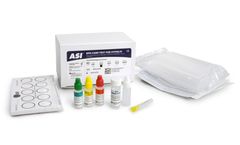
Manufactured by:NOWDiagnostics Inc. based inSpringdale, ARKANSAS (USA)
The ADEXUSDx Syphilis Test is a rapid immunochromatographic assay used for the qualitative detection of the presence or absence of antibodies to syphilis in human whole blood, plasma, or serum to aid in the diagnosis of Treponema ...
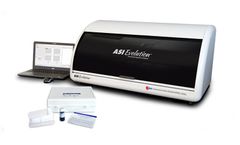
Manufactured by:Arlington Scientific, Inc. based inSpringville, UTAH (USA)
FDA Cleared For Diagnostic, Blood Donor and Cadaveric Tissue Screening; For over 37 years Arlington Scientific has been a pioneer in delivering new products and technologies to meet the growing demands of clinical diagnostics, blood donor and cadaveric tissue laboratories worldwide. The ASI Evolution® Automated Syphilis Analyzer was developed using the traditional ...
Manufactured by:Arlington Scientific, Inc. based inSpringville, UTAH (USA)
ASI’s nontreponemal RPR (Rapid Plasma Reagin) test screens for syphilis; a sexually transmitted disease caused by the bacterium Treponema pallidum (a spirochete bacteria). The disease is found worldwide and is easily treated if detected early. If left untreated it can be fatal and facilitate the transmission and acquisition of HIV.1 This test is FDA cleared for use in ...
Manufactured by:Chembio Diagnostic Systems, Inc. based inHauppauge, NEW YORK (USA)
A CE Marked rapid point-of-care test that simultaneously and separately detects treponemal and nontreponemal ...
Manufactured by:Jant Pharmacal Corporation based inEncino, CALIFORNIA (USA)
With the new universal slide-in rack system accommodating a variety of tube sizes, sample loading has become even easier and faster. This instrument is the world’s first fully automated system for nontreponemal RPR Syphilis testing. Simply load your reagents and up to 192 samples, then walk away. The AIX automates the processing, analysis, reporting and archival of results ...
Manufactured by:Chembio Diagnostic Systems, Inc. based inHauppauge, NEW YORK (USA)
An FDA Approved, dual rapid test for the detection of antibodies to HIV 1/2 and Treponema pallidum in fingerstick whole blood, venous whole blood, or plasma specimens. ...
Manufactured by:Gold Standard Diagnostics based inDavis, CALIFORNIA (USA)
The AIX1000 is the world’s first fully automated system for nontreponemal RPR Syphilis testing. Simply load your reagents and up to 192 samples, then walk away. The AIX automates the processing, analysis, reporting and archival of results for both RPR screens and titers. Process and Machine for Automated Agglutination Assays with Image Automated ...
Manufactured by:ZeptoMetrix - ANTYLIA Scientific based inBuffalo, NEW YORK (USA)
The syphilis positive donor plasmas used for this panel were obtained under informed consent and collected in US FDA licensed plasma centers. Donors were untreated and asymptomatic at the time of ...
Manufactured by:Arlington Scientific, Inc. based inSpringville, UTAH (USA)
Arlington Scientific’s Automated RPR Syphilis Test is FDA cleared for diagnostic, blood donor screening and cadaveric RPR testing, for use on the ASI Evolution® Automated Syphilis Analyzer. Nontreponemal tests, such as the ASI Automated RPR test, are more reliable indicators of untreated syphilis infections and are used to monitor ...
Manufactured by:Scimedx Corporation based inDover, NEW JERSEY (USA)
For the rapid detection and quantitative determination of syphilis in serum or plasma. The identification aids in the diagnosis of syphilis caused by the genus Treponema and provides epidemiological information on syphilis. 201-302/500 500 Test ...
Manufactured by:Genabio Diagnostics Inc. based inBedford, MASSACHUSETTS (USA)
The three major causes of Genital Ulcer Disease (GUD) in the United States are Herpes simplex virus, Treponema pallidum (syphilis) and Haemophilus ducreyi (chancroid). ...
Manufactured by:Cone Bioproducts based inSeguin, TEXAS (USA)
Individual donor units used in the preparation of this product have been tested and found to be negative or non-reactive for HBsAg, Anti-HIV I/II, Anti-HCV, HIV-1 RNA, HCV RNA. Donors were tested and found negative for Syphilis according to FDA ...
Manufactured by:Cone Bioproducts based inSeguin, TEXAS (USA)
Individual donor units used in the preparation of this product have been tested by FDA approved tests and found to be non-reactive for HBsAg, Anti-HIV I/II, Anti-HCV, HIV-1 RNA, HBV DNA and HCV RNA. Donors of human plasma units used in the making of this product were tested and found negative for Syphilis according to FDA ...
Manufactured by:Impladent Ltd. based inJamaica, NEW YORK (USA)
A donor serum sample is tested by a CLIA Certified Lab using FDA licensed screening tests and found to be negative or non-reactive for antibodies to human immunodeficiency virus type 1 and type 2 (anti-HIV 1 and anti-HIV 2), hepatitis B surface antigen (HbsAg), hepatitis B core antibody (HbcAb), antibodies to the hepatitis C virus (anti-HCV), and the human T-lymphotrophic virus type I antibody ...
Manufactured by:Cone Bioproducts based inSeguin, TEXAS (USA)
Cone Bioproducts’ Human Gamma Globulin is obtained via Cohn fractionation of human plasma. The lyophilized crystalline powder is soluble in water and is suitable for a wide range of applications in the life sciences, including but not limited to IVD Manufacturing. ...
Manufactured by:Cone Bioproducts based inSeguin, TEXAS (USA)
Part Number: 6107. Form/Appearance: Liquid, straw to amber, free of visible foreign matter or precipitates. Filtration: Available. Additives: None. Storage: ≤-20 °C. Expiration: 5 years from DOM. COVID-19 IgG Ab: Negative. pH: 7.1 - 7.5. Bioburden: < 100 ...
Manufactured by:Cone Bioproducts based inSeguin, TEXAS (USA)
Part Number: 5210. Format: Liquid. Source: Human Serum. Number of Levels: 3. Fill Volume: 0.5 mL. Features: Value. Assigned on Abaxis Piccolo. Storage: < -20 C. Open Vial/Reconstituted Refrigerated Stability: 14 days. Expiration: 18 months from date of manufacture. Analytes: Albumin, ALP, ALT, Amylase, AST, Direct Bilirubin, Total Bilirubin, BUN, CRP, Calcium, Bicarbonate, Chloride, Total ...
Manufactured by:Cone Bioproducts based inSeguin, TEXAS (USA)
Cone Bioproducts is a bulk manufacturer of human HDL cholesterol concentrate. HDL Cholesterol Concentrate is used as a component in quality controls, calibrators, interference studies and as a cell culture growth ...
Manufactured by:Cone Bioproducts based inSeguin, TEXAS (USA)
Cone Bioproducts is a bulk manufacturer of human HDL cholesterol concentrate. HDL Cholesterol Concentrate is used as a component in quality controls, calibrators, interference studies and as a cell culture growth ...
Manufactured by:Cone Bioproducts based inSeguin, TEXAS (USA)
Cone Bioproducts’ Folate, Vitamin B12 Depleted Human Serum Albumin is obtained via Cohn fractionation of Human plasma and is further processed to remove Folate and Vitamin B12. The lyophilized crystalline powder is soluble in water and is suitable for a wide range of applications in the life sciences, including but not limited to: IVD Manufacturing, Cell culture supplement, Biological ...