Refine by
Drug Safety Articles & Analysis
88 news found
Obtaining the Marketing Authorisation Application (MAA) is a key step in the commercialization of drugs in the EU, and also an important help to promote the global market expansion of pharmaceutical companies. ...
Ace Therapeutics Psychiatry Team is committed to supporting scientists in making groundbreaking scientific discoveries and developing new applications to accelerate drug discovery and the scientific diagnosis and treatment of antipsychotic disorders. ...
An Abbreviated New Drug Application (ANDA) is an application submitted to the U.S. FDA to demonstrate that a generic drug is equivalent to a previously approved Reference Listed Drug (RLD) in terms of safety, efficacy, and quality. The ANDA contains information used for the review and approval of a generic drug ...
Alfa Chemistry provides a wide variety of these intermediates which meet strict quality and safety requirements required for the drug development process. These intermediates (ammonium acids, organic acids, esters and heterocyclic compounds) allow us to innovate in drug synthesis, which in turn leads to more effective and precise ...
With the evolution of pharmaceutical industry over the past decades, compliance remains a key factor in bringing novel drugs to market. Proregulations, as a product safety and regulatory consulting firm with a long-standing core mission of "Value in Compliance", announces to offer a series of DMF document production and filing services, helping clients worldwide ...
The process of antipsychiatric drug development typically involves several stages, including identifying potential drug targets, conducting preclinical research to test the safety and efficacy of potential drug candidates, and then moving on to clinical trials to further evaluate the drug’s effectiveness ...
The applications of WGS are vast, impacting fields such as disease research, drug discovery, personalized medicine, food safety, agriculture, and pharmacy. Key advantages of WGS include single base-pair resolution, de novo sequencing and genome-wide mutation characterization, population evolution and phylogenetic studies, and its critical role in disease ...
CD ComputaBio, a pioneering provider of computational biology services, has recently introduced its advanced Computer-Aided Drug Design (CADD) services, revolutionizing the drug discovery landscape. ...
Toxicology is another cornerstone of its service suite, crucial for ensuring that new veterinary drugs meet strict safety standards. By conducting meticulous toxicological assessments, CD Formulation identifies potential side effects and toxicities, enabling the refinement of drug formulations before they reach the market. This proactive approach ...
The design of such drug carriers also helps to overcome some of the limitations of conventional drugs such as biostability, water solubility and drug distribution in the body. ...
First, the team would conduct extensive preclinical investigations to ascertain the safety and effectiveness of the oral thin films. Factors like drug release kinetics, absorption, metabolism, and possible toxicity during the preclinical stage would be assessed. ...
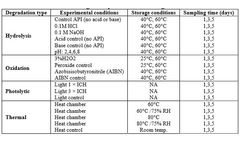
STEMart, a US-based provider of comprehensive services for all phases of medical device development, announced the launch of its new Force Degradation Services to help pharmaceutical and medical device companies evaluate the stability of their drug candidates and finished products under a variety of stress conditions, ensuring their safety and efficacy throughout ...
BySTEMart
In the field of pharmaceutical formulation, stability analysis is a critical aspect that ensures the safety and efficacy of drugs throughout their shelf life. CD formulation has introduced a series of stability analysis services as a prominent tool for researchers to understand the influence of various factors and tests on the stability of pharmaceutical ...
In the ever-evolving field of pharmaceutical research, the hunt for effective drugs often rests on the ability to understand and manipulate the solid-state properties of compounds. ...
CD Formulation understands the critical importance of selecting the right excipients, polymer matrices, and drug loading techniques to ensure optimal drug stability and release. ...
The results of these tests help researchers understand the evolutionary, ecological, and cognitive aspects of animal behavior, as well as explore mechanisms of diseases related to neurological dysfunction and animal models and evaluate the efficacy and safety of related drugs. ...
At present, anxiety and depression-related animal behavior tests are widely used in pharmacological research to evaluate the characteristics of candidate drugs for treating these diseases in rodent models. These tests can provide valuable insights into the potential clinical applications of compounds. ...
However, as with any medical product, it is crucial to assess the safety and potential toxicity of these novel drug delivery platforms. ...
Alfa Chemistry serves as an emerging pharmaceutical research organization with a mission to revolutionize the field of preclinical drug research with its wide range of comprehensive services. With a commitment to quality and excellence, Alfa Chemistry offers pharmacological analysis, drug safety evaluation, pharmacokinetic analysis, and ...
The newly introduced solutions are focused on long-acting controlled-release microspheres and phase-transition microneedle patches, to meet the growing demand for novel biopharmaceuticals and biosimilars, while at the same time, improving efficacy, safety, and convenience in drug delivery. Microsphere and microneedle technologies have been gaining increasing ...