Refine by
Femoral Artery Equipment & Supplies
53 equipment items found
Manufactured by:United Biologics, Inc. based inIrvine, CALIFORNIA (USA)
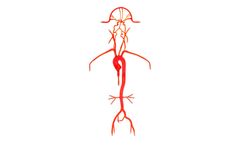
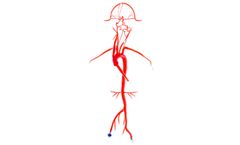
Our Neuro System Trainer extends from the femoral arteries to the pericallosals and M2, and A2 segments, and features six aneurysms of varying sizes. The left and right vertebrals are customized to simulate both a young and mature ...
Manufactured by:United Biologics, Inc. based inIrvine, CALIFORNIA (USA)
Our Neuro System Trainer extends from the femoral arteries to the pericallosals and A2, M2 segments, and features six aneurysms of varying sizes. The left and right vertebrals are customized to simulate both a young and mature adult. ...
Manufactured by:Viant Medical based inFoxborough, MASSACHUSETTS (USA)
Complex interventional devices. Steerable, balloon, and RF ablation catheters. Closure devices (femoral arterial and venous access). Introducers for transradial artery access. ...
Manufactured by:United Biologics, Inc. based inIrvine, CALIFORNIA (USA)
The Transcatheter Aortic Valve Replacement model is our transparent, versatile platform for training and product development. This vessel features two replaceable aortic valves, the left ventricle, and a full aorta down to the femoral arteries. Femoral access and modular heart valves allow for full simulation of transcatheter therapy scenarios ...
Manufactured by:United Biologics, Inc. based inIrvine, CALIFORNIA (USA)
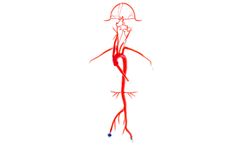
Our Neuro System Trainer extends from the femoral arteries to the pericallosals and A2, M2 segments, and features six aneurysms of varying sizes. The left and right vertebrals are customized to simulate both a young and mature adult. To secure your vessel during testing or demonstrations, we offer 3D/2D Stabilizing Platforms. ...
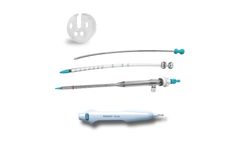
Manufactured by:Prytime Medical Devices, Inc. based inBoerne, TEXAS (USA)
The REBOA Convenience Set is compatible with all Prytime Medical™ REBOA catheters and provides the clinician with the components needed for common femoral artery access, REBOA placement, and external fixation of the ...
Manufactured by:Cordis based inMiami Lakes, FLORIDA (USA)
The EXOSEAL Vascular Closure Device is designed for a safe, simple, and secure close. The EXOSEAL VCD is indicated for femoral artery puncture site closure, reducing times to hemostasis and ambulation in patients who have undergone diagnostic or interventional catheterization procedures using a standard 5F, 6F, or 7F vascular sheath introducer with up to a 12-cm ...
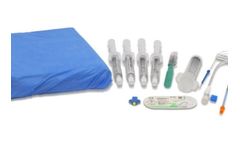
Manufactured by:Dispack Medical based inSarniç-Gaziemir, TURKEY
Step into the future of vascular procedures with confidence and precision using Dispack Medical’s Femoral Angiography Drape Pack. Specifically designed for femoral artery angiography, this Drape Pack ensures a sterile and controlled environment, empowering healthcare professionals in delivering optimal care during vascular ...
Manufactured by:Insightra Medical Inc, a Mundomedis Group Company based inClarksville, TENNESSEE (USA)
This ULTRA 7FR IAB catheter is a true 7Fr (including the wrapped balloon), so it will pass through all 7Fr sheaths. 7Fr means a 23% reduction on cross-sectional area (vs. a competitive 8Fr), which results in over 20% better distal blood flow in a 4.0mm femoral artery. ...
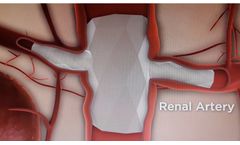
Manufactured by:Red Vascular Technology, LLC based inTampa, FLORIDA (USA)
Red Vascular’s non-modular, branched endoprosthesis represents an ideal solution for aortic aneurysmal disease with branch artery involvement. Its design and delivery through a single femoral artery site are superior to modular, fenestrated grafts currently used to treat complex aneurysms. Fenestration adds several complex ...
Manufactured by:MedChemExpress LLC (MCE) based inMonmouth Junction, NEW JERSEY (USA)
Niguldipine free base exhibits dose-dependent and sustained increases in coronary blood flow. Niguldipine free base also increases perfusion in the kidneys and femoral arteries, but the effect is temporary and to a lesser extent. The effect of Niguldipine free base on myocardial metabolism is not ...
Manufactured by:Viant Medical based inFoxborough, MASSACHUSETTS (USA)
Heart positioners, Aortic punches, Syringes, Arterial, femoral, and venous cannulae for extracorporeal circuits, Heart valve holders, Delivery ...
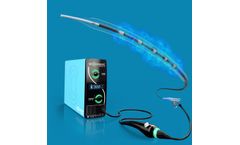
Manufactured by:Neurescue based inCopenhagen, DENMARK
It is the first computer-aided aortic occlusion catheter with pressure-controlled inflation and position feedback. Aortic balloon occlusion is a technique where a balloon is advanced into the descending aorta through the femoral artery and temporarily inflated, thereby redirecting blood flow to the heart and the brain. Our future goal is to enable the clinical ...
Manufactured by:Concept Medical Inc. based inTampa, FLORIDA (USA)
AN EXCELLENT ALTERNATIVE FOR REVASCULAZATION OF SFA AND BTK ARTERIES.MagicTouch PTA is intended to prevent re-narrowing of superficial femoral and popliteal arteries in patients with peripheral artery ...
Manufactured by:AllVascular Pty Ltd based inLeonards, AUSTRALIA
The Implant; Long term arterial implant anastomosed to either the femoral or axillary artery. Dual Dacron cuffs provide infection barrier and tissue anchorage. ePTFE vascular graft allows for end-to-side ...
Manufactured by:Nantah Capital One Pte Ltd based inSingapore, SINGAPORE
Affordable, portable, reusable wet lab box for evaluating all vascular anastomosis skills. Repair surrounding tissue and skin layer for a complete training experience. Use the pulsatile flow pump for arteries or continuous flow pump for veins to check for the quality of the repair. Cuts like skin. Dissects and sutures like real tissue. Use with any LifeLike or silicone ...
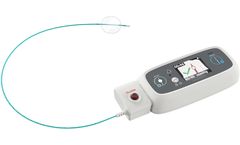
Manufactured by:Millar based inHouston, TEXAS (USA)
The Millar SPT-301 Non-Invasive Pulse Wave Tonometer is designed for a seamless evaluation of the cardiovascular system. Providing high-fidelity blood pressure waveforms through direct skin contact with the carotid, femoral, or radial arteries, it offers valuable insights into cardiovascular health. The device efficiently integrates with standard blood pressure ...
Manufactured by:Medinol based inTel Aviv, ISRAEL
ChampioNIR is a hybrid stent combining Nitinol alloy structure with an elastomer micro-fiber mesh. Fracture resistance during contraction, compression, torsion, extension and flexion. Outstanding radial support and conformability to the vessel natural form and motion. Unique delivery system for accurate stent deployment. Superior flexibility for better deliverability. ChampioNIR is the first ...
Manufactured by:Shockwave Medical Inc. based inSanta Clara, CALIFORNIA (USA)
Quicker Cycle Time With 2x Faster Pulsing. 2X Faster Pulsing: Shockwave M5+ delivers the same amount of energy quicker with the same safety and efficacy profile as Shockwave ...
Manufactured by:Newtech Medical Devices based inFaridabad, INDIA
In medical devices, a catheter is a thin tube extruded from medical grade materials (polyurethane & silicon) serving a broad range of functions. Catheters are medical devices that can be inserted in the body to perform a surgical ...