Refine by
In Vitro Diagnostic Use Suppliers Serving Kiribati
49 companies found

based inNew York City, NEW YORK (USA)
Led by a team of experts in the fields of life sciences, oncology, pathology, technology, machine learning, and healthcare, Paige strives to transform cancer diagnostics. We make it possible not only to provide additional information from digital ...

based inThessaloniki, GREECE
Hellabio is a European Company established in 1986, located in Business Incubator Thermi, Thessaloniki, Greece. We are active researcher and manufacturer of innovative biological reagents for in vitro diagnostic use. We are committed to meeting and ...

based inSomerset, NEW JERSEY (USA)
Even the most sophisticated, cutting-edge medical diagnostics aren’t of value unless we can get them to those who need them most. At Access Bio, we believe every life is precious, and every human on earth deserves the opportunity to live a healthy ...
The CareStart™ COVID-19 MDx RT-PCR is a real-time reverse transcription polymerase chain reaction (RT-PCR) test intended for the qualitative detection of nucleic acid from SARS-CoV-2 in respiratory specimens (such as nasopharyngeal, ...

based inLake Forest Park, WASHINGTON (USA)
IEH was founded in 2001 by Dalia Alfi and Mansour Samadpour, who left their positions at the University of Washington, School of Public Health, to start the company. It is a family-owned and operated company and has grown rapidly since 2001, both ...

based inHayward, CALIFORNIA (USA)
Novodiax is dedicated to advancing tissue-based diagnostics and immunoassays through innovation. Our signature technology platform is a proprietary polymer-based detection system. Based on this technology we have developed a series of highly ...
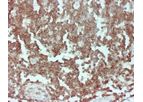
Cluster of Differentiation 45 (CD45) pHRP Anti-Human Staining Kit immunohistochemistry (IHC) reagent is for use on frozen and formalin-fixed-paraffin-embedded tissues. The CD45 antigen is a protein which was originally called Leukocyte Common ...

based inOmaha, NEBRASKA (USA)
ADS Biotec, along with its parent company ADSTEC (Japan), is a global leader in the development, manufacture and sale of automated instruments and consumables for use in cytogenetic, pathology and research laboratories. Our technology and services ...

based inAmherst, NEW HAMPSHIRE (USA)
BUHLMANN Diagnostics Corp (BDC), Amherst NH is the North American affiliate of BÜHLMANN Laboratories AG, a leading worldwide operative manufacturer of ELISA kits, RIA kits, lateral flow assay kits, flow cytometry assay kits and turbidimetric assays ...

based inSydney, AUSTRALIA
SpeeDx specializes in molecular diagnostic solutions that go beyond simple detection to offer comprehensive information for improved patient management. SpeeDx is a dynamic, rapidly growing company with a strong portfolio of technology at the ...
Detect COVID-19 causative coronavirus, SARS-CoV-2; Multiplex RT-qPCR test with dual targets (RdRp/ORF1ab) and RNA-based internal control in a single-well. Scalable solution supporting surge ...

based inSeoul, SOUTH KOREA
PROTIA Inc. aims to contribute to human health and well-being through finding new protein targets for disease prediction, early diagnosis, and development of new medicines. Based on our dedication for identification of bio-markers needed in early ...
ImmuneCheck IgG is an in vitro diagnostic kit for use in the semi-quantitative analysis of human IgG (hIgG) in human serum, plasma and whole blood samples using immunochromatography ...

based inUpper Coomera, AUSTRALIA
Bio Molecular Systems (BMS) is a biotechnology company that designs, manufactures and distributes instrumentation for the life sciences. Established in 2010 and comprising of the founders and senior personnel from the former Corbett Life Sciences ...
The world’s first magnetic induction cycler is now a registered medical device with CE-IVD and TGA approval. Mic IVD is manufactured under an ISO 13485:2016 Quality Management System. ...

based inManchester, UNITED KINGDOM
Yourgene Health is a leading international molecular diagnostics group with an integrated portfolio of technologies and services to enable genomic medicine. Our mission to enable scientific advances to positively impact human health remains at the ...
The Yourgene QST*R Pregnancy Loss assay is for the routine in vitro quantitative diagnosis of the six most common autosomal trisomies associated with pregnancy ...

based inIrvine, CALIFORNIA (USA)
MP Biomedicals is a worldwide corporation developing, manufacturing and distributing products for the life science, in vitro diagnostics (IVD), fine chemicals and dosimetry markets. Through our global operational network, we provide research and ...
The Neonatal 17OHP ELISA Kit is intended for the measurement of 17-alpha hydroxyprogesterone (17OHP) in a whole blood ...

based inPoway, CALIFORNIA (USA)
Located in San Diego County, California, an area well known for biotechnology and scientific discovery, CTK Biotech develops and manufactures innovative immunodiagnostic tools and point of care diagnostic test kits for the IVD community worldwide. ...
The RaFIA Immunofluorescence Analyzer is a fluorescence immunoassay analyzing instrument intended for use by healthcare professionals to aid in the diagnosis of conditions such as cardiovascular disease, pregnancy, infection, diabetes, renal injury ...

based inBelgrade, SERBIA
Yunycom d.o.o. Belgrade was founded in 1990. as a private company and has been developing their business in the area of import, export and distribution of medical equipment, diagnostic materials and cosmetic products and services. Yunycom’s primary ...

based inYork, UNITED KINGDOM
Abingdon Health is a world leading developer and manufacturer of high-quality rapid lateral flow tests across all industry sectors, including healthcare, animal health and plant health. We take projects from initial concept through to routine and ...

based inKAARINA, FINLAND
Labmaster Oy Ltd. is a company engaged in the design, development, and manufacturing of various products, ensuring they meet specified criteria through proper storage, usage, and transportation guidelines. They emphasize adherence to instructions ...
The Labmaster LUCIA™ MxA Kit is a sophisticated in vitro diagnostic tool used for the quantitative assessment of the Myxovirus resistance protein A (MxA) in whole blood samples. This ...

based inNanjing, CHINA
Founded in 2002, Jiji Biotech Co., Ltd.is a medical and health industry chain that integrates independent research and development, large-scale production and professional marketing. It has many subsidiaries and offices at home and abroad. the ...
Can be used as a aid in identifying individuals with an adaptive immune response to SARS-CoV-2, indicating recent or prior infection, it should not be used to diagnose acute SARS-CoV-2 infection. ...

based inXiamen, CHINA
Amoy Diagnostics Co., Ltd. (AmoyDx) is an R&D based diagnostic company, focusing on molecular diagnostics for oncology precision medicine. With completely independent intellectual property rights of ADx-ARMS®, Super-ARMS®, ddCapture® and ADx-HANDLE® ...
Lung cancer is one of the most common malignant tumor, and 80~85% of lung cancers are non-small cell lung cancer (NSCLC). There are many driver mutations in NSCLC. The frequency of mutations in NSCLC for EGFR, HER2, KRAS and BRAF genes are ...

based inSalt Lake City, UTAH (USA)
What we do is driven by what we believe. Which is helping create a better quality of life on a large scale. That’s why we are making our revolutionary PCR technology available at cost-efficient pricing around the world. No matter your geography and ...
The Logix Smart™ Coronavirus Disease 2019 (COVID-19) Test kit is an in vitro diagnostic test that uses our patented CoPrimer™ technology for the qualitative detection of the RNA from ...

based inUdine, ITALY
Ulisse BioMed is an Italian healthcare biotech company with corporate offices in Udine, Italy and with R&D and Manufacturing labs in Trieste, Italy. Ulisse BioMed is creating novel, innovative and transformative diagnostic platforms and products. ...
The assay is intended for the qualitative detection of RNA from SARS-CoV-2 in clinician-collected nasopharyngeal swab specimens from patients with signs and symptoms suggestive of COVID-19 (e.g., fever and/or symptoms of acute respiratory ...