- Home
- Companies
- faroe islands
- vascular access
Show results for
Refine by
Vascular Access Suppliers Serving Faroe Islands
57 companies found

based inDeerfield, ILLINOIS (USA)
Baxter was founded in 1931, however our history doesn't have one starting point. Our commitment to addressing unmet needs was built on the shoulders of healthcare pioneers whose work resulted in first-of-its-kind innovations in IVs, renal care, ...

based inViadana, ITALY
Delta Med was founded in 1993 with the aim of developing and manufacturing safe and high quality medical devices. With more than twenty years of experience behind us, over time we have become a reference point in health and safety, and privileged ...
The range of MICROCATH long cannulae is one of the most important international innovations regarding vascular accesses. Made of Pebax and suitable for use within 29 days, sterile venous catheters are radiopaque and ...

based inSalt Lake City, UTAH (USA)
At Health Line, our mission is to be one of the world’s leading manufacturers of vascular access, infusion therapy, and infection control products. We are committed to upholding strong quality management and regulatory systems to meet the demands of ...
The ORION™ II CT Central Venous Catheters are power injectable and intended for short-term infusion therapy. Available in a variety of sizes and lengths, the ORION™ II has an indwelling central line indicated for pressure injection of ...

based inCantù (CO), ITALY
Bioengineering Laboratories (BEL) has been conceived as a young, streamlined and multi-faceted organization able to provide technical knowledge and a twenty year experience in designing, testing and producing innovative medical devices in the ...

based inCalgary, ALBERTA (CANADA)
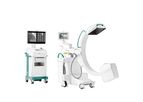
Circle improves patient outcome by revolutionizing the cardiovascular healthcare industry through innovation, and enabling healthcare providers to complete effective and precise analysis. The Global Leader in Cardiac Imaging Solutions. Physicians ...
cvi42 for fast and efficient planning of structural heart ...

based inCoralville, IOWA (USA)
For more than 35 years, CIVCO Medical Solutions has been committed to making image-guided procedures safer through the design, manufacturing, and marketing of medical products. Our products are trusted by physicians and clinicians in radiology, ...

based inMississauga, ONTARIO (CANADA)
Covalon Technologies Ltd. (Covalon) develops and commercializes proprietary technologies for major medical device manufacturers. Our partners include the top medical device companies, leaders in urology, wound care, vascular access, venture capital, ...
A highly versatile coating, CovaCoat is a photochemical process, that enables permanent covalent attachment of the coating to a wide variety of medical devices. Each formulation is specifically tailored to the device, and can be engineered to ...

based inLeonards, AUSTRALIA
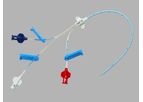
AllVascular is an Australian medical device company based in Sydney. The Company’s lead product is a novel vascular access system called the AVAS. The AVAS is unique as it can facilitate treatments that require frequent vascular access such as the ...
The Implant; Long term arterial implant anastomosed to either the femoral or axillary artery. Dual Dacron cuffs provide infection barrier and tissue anchorage. ePTFE vascular graft allows for end-to-side ...

based inWaldbuttelbrunn, GERMANY
In 2000, TauroPharm GmbH was founded as a German company specialising in medical devices with antimicrobial efficacy. We have since developed a variety of innovative products and built an international network. As TauroLock solutions have been ...
If you are looking for a catheter lock solution with both an antimicrobial and anticoagulant effect, we recommend TauroLock™. On this site, you can find detailed information about the product, its active ingredients, and ...

based inEindhoven, NETHERLANDS
Xeltis is a clinical-stage medical device company pioneering a restorative approach in cardiovascular therapy. RestoreX™, Xeltis technology platform, is the world’s first polymer-based technology designed to enable natural restoration of ...
The aXess graft is a restorative, synthetic, electrospun blood vessel for arteriovenous hemodialyisis access, overtime turning into a living vessel made of patient’s own ...

based inParis, FRANCE
emka TECHNOLOGIES, now joined with SCIREQ, has been providing integrated systems for preclinical physiology, pharmacology & toxicology research for over 25 years. Our hardware and software are used daily, validated by major pharma companies and CROs ...

based inMinneapolis, MINNESOTA (USA)
At Medtronic, we believe in the power of medical technology to improve lives. Seven decades ago, our co-founder invented the battery-powered pacemaker. Today, we are among the largest medical device companies in the world. With operations in 150 ...
The Crescent™* catheter is the first FDA-cleared, jugular dual lumen long-term ECMO catheter. It allows for more accurate placement with just one cannulation site, delivers enhanced flow dynamics, and helps maintain optimal flow† once ...

based inIrvine, CALIFORNIA (USA)
JenaValve is a clinical-stage medical device company developing the first transcatheter heart valve technology that is uniquely designed for the minimally invasive treatment of both severe aortic regurgitation and stenosis. JenaValve is passionate ...
The JenaValve Transfemoral Delivery Catheter is designed to safely treat patients while delivering consistent, reproducible performance for physicians. The system features the ...

based inCarlsbad, CALIFORNIA (USA)
CeloNova BioSciences, Inc. is developer and manufacturer of a portfolio of novel surfaced coated technologies, including the proprietary Polyzene-F nanocoating. This next-generation nanocoating is the result of years of rigorous scientific research ...

based inLabège, FRANCE
Created in 1985 by Pierre Montoriol, Hemodia head office is located in Toulouse, in the south west of France. The company is known for its expertise in the design, production and marketing of single-use medical devices. Hemodia is a key player on ...
Care kits for: cannulation and line insertion, immediate or deferred infusion connection, medication reconstitution for drug delivery, filling of infusion devices, disconnection, needle withdrawing, maintaining the permeability of the ...

based inDurham, NORTH CAROLINA (USA)
Humacyte, Inc. (Nasdaq: HUMA) is developing a disruptive biotechnology platform to deliver universally implantable, bioengineered human tissues and organs designed to improve the lives of patients and transform the practice of medicine. Humacyte ...
We are initially using our technology to bioengineer and manufacture human acellular vessels, or HAVs, in varying sizes to address the significant unmet needs across multiple potential indications in vascular repair, reconstruction ...

based inBloomington, INDIANA (USA)
There are common themes across every medical specialty we support: patients want to get better and doctors want simpler, more effective options. That’s why we work hard to improve our minimally invasive medical devices and the way they’re delivered ...
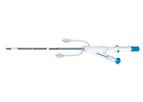
The central venous catheter is designed for treatment of critically ill patients and is suggested for: 1. Continuous or intermittent drug infusions; 2. Central venous blood pressure monitoring (CVP); 3. Acute hyperalimentation; 4. Blood sampling; ...

based inAlpharetta, GEORGIA (US) (USA)
Founded by Fred L. Aycock in 1992, Remington Medical Inc. specializes in medical devices. The maintenance, time, and money put into cleaning and sterilizing medical devices led us to develop some of the first disposable cables, some still in use ...

based inSeattle, WASHINGTON (USA)
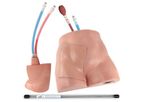
Since 1994, Simulab has been committed to providing medical simulators and task trainers to the medical education community. By collaborating with leading educators worldwide, leveraging its years of experience, and bringing a specialized knowledge ...
Simulab's FemoraLineMan Training Package is an ultrasound-compatible task trainer that offers an effective training solution for central venous or arterial access using the femoral site. This femoral line trainer uses the same patented ...

based inSincan, TURKEY
As Istem Medikal employees, together with our business partners, we serve our customers by manufacturing and supplying innovative, reliable and environment friendly medical devices. We diligently value our brand and its benefit to society. Istem ...
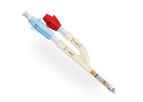
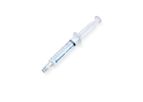
Prefilled Syringe with Sterile 0,9% Sodium Chloride Solution. SENSOFLUSH is specifically designed for the flushing of in-situ vascular access devices in order to maintain patency and reduce the risk of ...