Refine by
Drug Stability Articles & Analysis
38 news found
Challenges in Pharmaceutical Development Improving the dissolution, solubility, and bioavailability of poorly soluble drugs (BCS Class 2) remains a significant challenge. Amorphous Solid Dispersions (ASDs) are a promising solution, stabilizing drugs in their amorphous form using polymers and low-molecular-weight excipients. ...
Transdermal Delivery: Overcoming Natural Barriers Traditional transdermal drug delivery has struggled with the skin’s natural barrier, the stratum corneum, which limits effective drug absorption. ...
“Whether customers are trying to develop a new drug or seeking to improve the release characteristics of an existing drug, our lyophilization and flash-release technology are there to help,” said Protheragen-ING’s Chief Scientist. Over the past decade, the use of the freeze-drying process in pharmaceuticals, biopharmaceuticals and nanoderived ...
Among the 15 approved ADCs globally, notable therapies like Trodelvy and Zynlonta utilize PEG linkers to enhance drug stability and targeting. The benefits of incorporating PEGs into ADC formulations are significant, including improved solubility, prolonged circulation time, and reduced immunogenicity, all of which contribute to more effective cancer treatments. ...
This strategic move is aimed at addressing the rising global demand for Lipid Nanoparticles (LNPs), which are vital for nucleic acid drug delivery, especially in mRNA-based therapies and vaccines. Lipid Nanoparticles have become a groundbreaking delivery mechanism within the biopharmaceutical sector. ...
The Y-Shape PEG NHS is particularly valuable in drug development and therapeutic protein formulation. It enhances drug stability, prolongs circulation time, and reduces immunogenicity, making it an essential tool for creating more effective and safer biologic drugs. ...
CD Formulation is making waves in the pharmaceutical industry with its innovative drug delivery technologies. With a focus on sustained and controlled release formulations, as well as liquid, semi-solid, injectable, and nanoformulations, CD Formulation is at the forefront of the industry's efforts to enhance drug efficacy. ...
Typically, the structures of such polymers are designed to reduce their dispersion in solution, thereby improving their stability and controllability. They’re widely used as drug carriers mainly because of their ability to efficiently carry and release drug molecules, thereby improving drug bioavailability and therapeutic ...
CD Formulation, a leading innovator in drug delivery technologies, has recently introduced a series of groundbreaking microneedle patch innovations that promise to revolutionize the way drugs are administered. ...
STEMart, a US-based provider of comprehensive services for all phases of medical device development, announced the launch of its new Force Degradation Services to help pharmaceutical and medical device companies evaluate the stability of their drug candidates and finished products under a variety of stress conditions, ensuring their safety and efficacy throughout ...
BySTEMart
CD Formulation's cutting-edge research and development team has successfully created micro-reservoir systems that offer precise drug release kinetics. By manipulating key factors such as reservoir size, drug loading, and polymer selection, the team has achieved remarkable control over drug release rates. ...
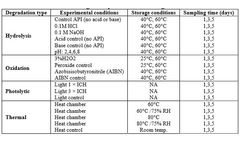
In the field of pharmaceutical formulation, stability analysis is a critical aspect that ensures the safety and efficacy of drugs throughout their shelf life. ...
These advanced systems offer unique benefits, including enhanced drug stability, sustained release, and targeted delivery. However, the successful formulation and optimization of these delivery systems pose significant challenges that require expertise and advanced technologies. ...
CD Formulation's transdermal patch drug delivery system services are designed to optimize formulations, ensuring the efficient and precise delivery of drugs across the skin barrier. ...
These new technologies, including drug half-life extension technologies, injection site drug reservoir technologies, and drug physicochemical enhancement technologies, offer new possibilities in the treatment of many chronic diseases. ...
The newly introduced solutions are focused on long-acting controlled-release microspheres and phase-transition microneedle patches, to meet the growing demand for novel biopharmaceuticals and biosimilars, while at the same time, improving efficacy, safety, and convenience in drug delivery. Microsphere and microneedle technologies have been gaining increasing popularity in ...
“In the beginning, people may view pharmaceutical analysis as analytical chemistry dealing with drug substances and pharmaceutical products, but in fact, analytical chemists are involved in many more studies, including the analysis of the chiral purity and quality of pharmaceutical formulations, drug quality control as well as pharmacokinetic ...
Although the drugs themselves are complicated, it is urgent to develop techniques to characterize glycosylation in order to better evaluate their effects. One of the most significant post-translational modifications during protein characterization is glycosylation. It affects the drug's biological activity, serum half-life, stability, and ...
The ARAI autoinjector has been developed by Aktiv with the support of BARDA and the Department of Defense (DoD) as a next generation autoinjector platform to address the shortfalls of previous autoinjectors, by improving usability, reliability, robustness, and drug stability. ARAI is a versatile platform that can deliver up to 5 ml dose volumes, perform ...
Broomfield Colorado 6/16/2022. Aktiv Pharma Group (Aktiv) announced today the acceptance of its novel glass-free, film-based flexible primary drug container (PDC) technology for sterile injectables named ARCH, into the US Food and Drug Administration (FDA) Emerging Technology Program (ETP). The ETP is a collaborative effort which supports the FDA’s mission to facilitate modernization in ...